A Taste of What Our PIs Are Working on...
Now for Some Details...
Click “MORE” Under Each Profile to Get ALL the Details
Anita E. Autry, Ph.D.
Assistant Professor, Neuroscience
(neuroscience category)

Molecular and circuit underpinnings of social behavior
Our laboratory is focused on uncovering and dissecting neural circuits that control social behaviors and understanding how these circuits are regulated under physiological and pathological conditions. Specifically, we study parental behavior which is essential for the health and survival of offspring, as well as infant-directed aggression and other behaviors associated with parenting. The research questions center around (1) how stress affects the function of circuits controlling parental behaviors (2) how circuits that mediate stress responses interact over time and (3) how stress circuits impact feeding behavior and body composition, particularly in lactating females.
Selected Publications
Kohl, J., Babayan, B. M., Rubinstein, N.D., Autry, A. E., Marin-Rodriguez, B., Kapoor, V., Miyamaishi, K., Zweifel, L. S., Luo, L., Uchida, N., Dulac, C. (2018). Functional circuit architecture underlying parental behavior. Nature. 556 (7701) 326–331.
Kohl, J.K.*, Autry A.E.*, Dulac, C. (2017). The Neurobiology of Parenting: A Neural Circuit Perspective. Bioessays, 39(1) 1–11.
Adachi, M.*, Autry, A.E.*, Maghoub, M., Suzuki, K., Monteggia, L.M. (2017). TrkB Signaling in Dorsal Raphe Nucleus is Essential for Antidepressant Efficacy and Normal Aggression Behavior. Neuropsychopharmacology, 42(4) 886–894.
Autry, A.E. (2016). Neurobiology of Chronic Social Defeat Stress: Role of Brain-Derived Neurotrophic Factor Signaling in the Nucleus Accumbens. Biological Psychiatry, 80(6) 39–40.
Morris, M.J., Na, E.S., Autry, A.E., Monteggia, L.M. (2016). Impact of DNMT1 and DNMT3a forebrain knockout on depressive- and anxiety like behavior in mice. Neurobiology of Learning and Memory, 135; 139–145.
Renier, N., Adams, E., Kirst, C., Wu, Z., Azevedo, R., Kohl, J., Autry, A.E., Kadiri, L., Venkataraju, K.U., Zhou, Y., Wang, V.X., Tang, C.Y., Olsen, O., Dulac, C., Osten, P., Tessier-Lavigne, M. (2016). Mapping of brain activity by automated volume analysis of immediate early genes. Cell, 165 (7) 1–14.
Wu, Z., Autry, A.E., Bergan, J.F., Watabe-Uchida, M., Dulac, C.G. (2014). Galanin neurons in the medial preoptic area govern parental behavior. Nature, 509 (7500) 325–330.
Nosyreva, E., Autry, A.E., Kavalali, E.T., Monteggia, L.M. (2014). Age dependence of the rapid antidepressant and synaptic effects of acute NMDA receptor blockade. Frontiers in Molecular Neuroscience, 94; 1–7.
Nosyreva, E., Szabla, K., Autry, A.E., Ryazanov, A.G., Monteggia, L.M., Kavalali, E.T. (2013). Acute suppression of spontaneous neurotransmission drives synaptic potentiation. Journal of Neuroscience, 33(16) 6990–7002.
Benekareddy, M., Nair, A.R., Dias, B.G., Suri, D., Autry, A.E., Monteggia, L.M., Vaidya, V.A. (2013). Induction of the plasticity-associated immediate early gene Arc by stress and hallucinogens: role of brain-derived neurotrophic factor. International Journal of Neuropsychopharmacology, 16(2) 405-15.
Costa Dobrenis, Ph.D.
Assistant Professor, Neuroscience
(neuroscience category)

Neurodegenerative lysosomal storage disease
Our principal interests lie in the pathogenesis and therapy of neurodegenerative diseases, and in the fields of ganglioside and microglial biology. We have contributed to the characterization of animal models of neuronal lysosomal storage diseases including Tay-Sachs/Sandhoff disease, Niemann Pick C (NPC) disease, mucopolysaccharidosis (MPS) IIIA, and mucolipidosis IV, and more recently to understanding the pathogenesis of Christianson Syndrome, a sodium/hydrogen exchanger deficiency that displays features of storage disorders. Much of our work has been directed at developing rational therapeutic strategies for these and related genetic diseases which affect the central nervous system (CNS) in a global manner. The goal here is to find ways to effectively replace the missing protein, or compensate for its function, within cells throughout the CNS. This entails overcoming challenges such as the blood brain barrier, and developing strategies that enhance neuronal uptake of therapeutic compounds.
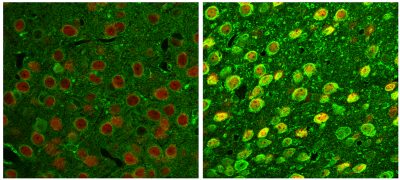
Immunofluorescent staining of neocortex from wild type (left) and Niemann Pick C disease mice showing significant upregulation and altered distribution of an ion channel (green) in neurons (NeuN-positive; red) not previously studied in this disease, but implicated by gene expression studies by the lab also suggesting its correction by cyclodextrin therapy of disease mice.
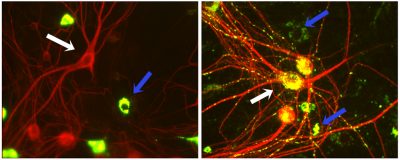
Neuronal cultures incubated with a soluble green reporter protein as is (left), or coupled to a peptide (right), developed by the lab, to enhance neuronal endocytic uptake and delivery to lysosomes for enzyme replacement therapy for lysosomal storage diseases. Uptake of the fluorescent protein (green) without the peptide (left) by neurons (red; positive for MAP2-immunostaining; e.g. white arrows) is virtually undetectable in contrast to more endocytically active glia (MAP2-negative; blue arrows). With coupled peptide (right), neuronal uptake is dramatically enhanced.
One of our ongoing projects in this regard is the development of fusion genes of hexosaminidase, the enzyme deficient in Tay Sachs disease, and peptide sequences related to the atoxic fragment of tetanus toxin. Due to characteristics of the latter, the encoded chimeric proteins have properties allowing circumvention of the blood brain barrier, increased neuronal endocytotic uptake into the lysosomal compartment, and transneuronal transsynaptic trafficking for wider dissemination of the needed protein. Additional projects include: exploring the efficacy and delineating the mechanisms of action of small molecule therapies such as miglustat, related analogs, and cyclodextrin for storage diseases, now seeing human application, in cellular, biochemical, gene expression and behavioral assays; and investigation of novel contact-mediated mechanisms of neuronal-microglial lysosomal enzyme transfer for effective hematopoietic stem cell replacement CNS therapy. Furthermore we continue to be engaged in studies examining the role and expression patterns of gangliosides and microglia in development and neuropathology. The lab utilizes techniques ranging from molecular recombinant work to animal behavioral assays with extensive experience in: cell culture preparations of all the major CNS cell types; gangliosides and lysosomal enzyme biochemistry; vital and fixed specimen histologic and immunocytochemical techniques; modern fluorescent techniques for monitoring organellar or biochemical activities in living cells; and a wide range of high resolution imaging and image analysis techniques.
Selected Publications
Boudewyn, L.C., Sikora, J., Kuchar, L., Ledvinova, J., Grishchuk, Y., Wang, S., Dobrenis, K., Walkley, S.U. N-butyl-deoxynojirimycin delays motor deficits, cerebellar microgliosis and Purkinje cell loss in a mouse model of mucolipidosis type IV. Neurobiol. Disease, 105:257-270, 2017.
Trilck, M., Peter, F., Zheng, C., Frank, M., Dobrenis, K., Mascher, H., Rolfs, A., Frech, M.J. Diversity of glycosphingolipid GM2 and cholesterol accumulation in NPC1 patient-specific iPSC-derived neurons. Brain Res. 1657:52-61, 2017.
Yang, D.-S., Stavrides, P., Kumar, K., Jiang, Y, Mohan, PS., Ohno, M., Dobrenis, K., Davidson, C.D., Saito, M, Pawlik, M., Huo, C., Walkley, S.U., Nixon, R.A. Cyclodextrin has conflicting actions on autophagy flux in vivo in brains of normal and Alzheimer model mice. Human Mole. Genet. 26:843-859, 2017.
Davidson, C., Fishman, YI, Puskas I., Szeman, J., Sohajda, T., McCauliff, L.A., Sikora, J., Storch, J., Vanier M.T., Szente, L., Walkley, S.U., Dobrenis, K. Efficacy and ototoxicity of different cyclodextrins in Niemann-Pick C disease. Ann Clin Transl Neurol. 3:366-380, 2016.
Saito, M., Wu, G., Hui, M., Masiello, K., Dobrenis, K., Ledeen, R.W., Saito, M. Ganglioside accumulation in activated glia in the developing brain: comparison between WT and GalNacT KO mice. J. Lipid Res. 56:1434-1448, 2015.
Farfel-Becker, T., Vitner, E.B., Kelly, S.L., Bame, J.R., Duan, J., Shinder, V., Merrill Jr, A.F., Dobrenis, K., Futerman, A.H. Neuronal accumulation of glucosylceramide in a mouse model of neuronopathic Gaucher disease leads to neurodegeneration. Human Molec. Genet 23:843-854, 2014.
Micsenyi, M.C., Sikora, J., Stephney, G., Dobrenis, K., Walkley, S.U. Lysosomal membrane permeability stimulates protein aggregate formation in neurons of a lysosomal disease. J. Neurosci. 33:10815-10827, 2013.
Erblich, B., Zhu, L., Etgen, A., Dobrenis, K., Pollard, J.W. Absence of colony stimulating factor-1 receptor signaling results in loss of microglia, disrupted brain development and olfactory deficits. PloS One, 2011. 2011;6(10):e26317. Epub 2011 Oct 27.
Stromme, P., Dobrenis, K., Sillitoe, R.V., Gulinello, M., Ali, N.F., Davidson, C., Micsenyi, M.C., Stephney, G., Ellevog, L., Klunglund, A., Walkley, S.U. X-linked Angelman-like syndrome caused by Slc9a6 knockout in mice exhibits evidence of endosomal-lysosomal dysfunction. Brain 134:3369-83, 2011.
Gulinello, M., Chen, F., Dobrenis, K. Early deficits in motor coordination, cognitive function and anxiety-like behavior in a mouse model of the neurodegenerative lysosomal storage disorder, Sandhoff disease. Behav. Brain Res. 193:315-319, 2008.
Tiago Goncalves, Ph.D.
Assistant Professor, Neuroscience
(neuroscience category)

How experience and neuronal activity contribute to the differentiation; neuronal maturation process in disease
The dentate gyrus (DG) is the main input region of the hippocampus. As such, it plays a crucial role in hippocampal function, including learning and memory. Additionally, the DG is one of only two regions of the mammalian brain that continuously add new neurons through adulthood, and there is increasing evidence that these adult-born neurons play a role in specific learning tasks: particularly, the ability to distinguish between similar memories. Adult neurogenesis constitutes an unusual mode of plasticity in the brain, with specific stimuli such as environmental enrichment and voluntary exercise modulating the number of neurons integrating DG networks. In addition, adult-born neurons have been used to study neuronal development, and defects in neurogenesis have been associated with several human neurological and psychiatric diseases. The main focus of my research interests is to understand how enrichment and activity regulate the proliferation of adult neural stem cells and shape nascent adult-born neurons during their integration into DG circuits.
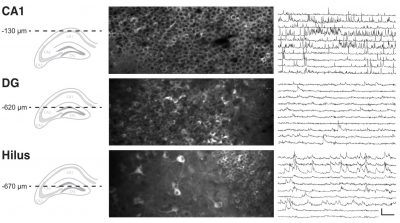
In vivo Ca2+ imaging of different hippocampal areas 3-photon images and Ca2+ traces of neurons expressing a genetically encoded Ca2+ sensor in CA1, DG granule layer and hilus/sub-granular layer. Note differences in morphology and activity patterns betwen cells in these layers. Scale bars: 100% ∆F/F 20 s.
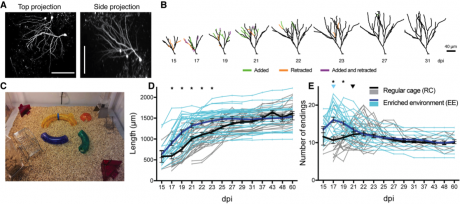
Exposure to an enriched enviroment (EE) leads to faster dendritic growth and earlier pruning (A) In vivo 2-photon images of RV-GFP labeled cells in the DG imaged 60 days post-infection (dpi). Scale bar = 100 µm. (B) Representative reconstructions of the dendrites of two newborn DGCs, branches added and retracted between imaging time points are highlighted. (C) EE cage is large (91×91) cm) and contains running wheels. (D) Individual and mean (bold) plot of dendrite length. (D) Branching of EE dendrites peaks earlier and maximum number of endings is higher than in RC dendrites (adapted from Gonçalves et al. 2016)
Experience and the activity it elicits are powerful modulators of neuronal development. As the brain matures it undergoes several critical periods where specific behavioral experiences (for example environmental enrichment and exercise) are needed for correct circuit formation. In the adult brain, behavioral experience has been shown to influence the proliferation of neural stem cells in the subgranular layer of the DG, their differentiation into glia and neurons, the maturation of these adult-born neurons, and their connectivity and incorporation into hippocampal circuits. Yet many aspects of the effects of experience on adult neurogenesis and hippocampus networks remain poorly understood. In our lab we study the effects of activity and experience at different phases of neuronal development ranging from the neural precursor stage to full maturity. The ability to track individual cells over their maturation period is crucial for our experiments. We use in vivo 2-photon imaging as a powerful tool to study of neuronal development since it allows for the recurrent probing of individual cells and circuits while keeping sensory input, neuronal activity patterns, and the physiological environment intact.
Selected Publications
Mansour A.F., Gonçalves J.T., Bloyd C.W., Li H., Fernandes S., Quang D., Johnston S., Parylak S.L., Jin X, Gage F.H., An in vivo model of functional and vascularized human brain organoids, Nat. Biotechnol. , doi:10.1038/nbt.4127 (2018)
O’Donnell C., Gonçalves J.T., Portera-Cailliau C., Sejnowski T.J., Beyond excitation/inhibition imbalance in multidimensional models of neural circuit changes in brain disorders, eLife, 6:e26724 (2017)
O’Donnell C., Gonçalves J.T., Whiteley N., Portera-Cailliau C., Sejnowski T.J., The population tracking model: A simple, scalable statistical model for neural population data, Neural Computation, 29, 50–93 (2017)
Gonçalves J. T. *, Schafer S.T. * (equal contribution), Gage F.H., Adult neurogenesis in the hippocampus: from stem cells to behavior, Cell, 167, 897–914 (2016) review article
Gonçalves J.T., Bloyd C.W., Shtrahman M, Johnston S.T., Schafer S.T., Parylak S.L., Thanh T., Chang T., Gage F.H., In vivo imaging of dendritic pruning in dentate granule cells, Nat. Neurosci., 19(6), 788–791 (2016) (journal cover)
Poo M.M., Pignatelli M., Ryan T.J., Tonegawa S., Bonhoeffer T., Martin K.C., Rudenko A., Tsai L., Tsien R.W., Fishell G., Mullins C., Gonçalves J.T., Shtrahman M., Johnston S.T., Gage F.H., Dan Y., Long J. Buzsáki G., Stevens C., What is memory? The present state of the engram, BMC Biology, 14(1):40 (2016) opinion article
Mertens J., Paquola A. C. M., Ku M., Hatch E., Böhnke L., Ladjevardi S., McGrath S., Campbell B., Lee H., Herdy J. R., Gonçalves J. T., Toda T., Kim Y., Winkler J., Yao J., Hetzer M. and Gage F. H., Directly reprogrammed human neurons retain age-related transcriptomic signatures and reveal age-related nucleocytoplasmic defects, Cell Stem Cell, 17, 1–14 (2015)
Gonçalves J.T., Anstey J.E., Golshani P., Portera-Cailliau C., Circuit level defects in the developing neocortex of fragile X mice, Nat. Neurosci., 16, 903–09 (2013)
(equal contribution) Cheng A.*, Gonçalves J.T. *, Golshani P., Arisaka K., Portera-Cailliau C., Spatio-temporal excitation-emission multiplexing for multiple beam 2-photon calcium imaging in deep tissue, Nat. Methods., 8, 139–42 (2011)
Golshani P., Gonçalves J.T., Khoshkhoo S., Mostany R., Smirnakis S., Portera-Cailliau C., Internally mediated developmental desynchronization of neocortical network activity, J. Neurosci., 29, 10890–10899 (2009)