Research, Research, More Research...
Kamran Khodakhah Ph.D.
Department Chair, Dominick P. Purpura Department of Neuroscience
Behavioral/Cellular Molecular/Clinical

The goal of our laboratory is to understand the role of the cerebellum and basal ganglia in motor function and in movement disorders. Of particular interest to us is not only to understand the role of each structure in motor control, but also the manner in which they communicate to coordinate and complement each other. We approach these questions from both basic science and clinical perspectives. We use a combination of techniques, from behavioral studies to imaging and two photon microscopy and electrophysiology (both in vitro and in vivo). Our studies take advantage of normal and transgenic animal models.
Selected Publications
Calderon DP, Fremont R, Kraenzlin F, Khodakhah K. (2011) The neural substrates of rapid-onset Dystonia-Parkinsonism. Nat Neurosci. Mar;14(3):357–65.
Dizon MJ, Khodakhah. (2011) The role of interneurons in shaping Purkinje cell responses in the cerebellar cortex.K. J Neurosci. Jul 20;31(29):10463–73.
Alviña K, Khodakhah K. (2010) The therapeutic mode of action of 4-aminopyridine in cerebellar ataxia. J Neurosci. 26;30(21):7258–68.
Walter JT, Khodakhah K. (2009) The advantages of linear information processing for cerebellar computation. Proc Natl Acad Sci U S A. 106(11):4471–6
Alviña K, Walter JT, Kohn A, Ellis-Davies G, Khodakhah K. (2008) Questioning the role of rebound firing in the cerebellum. Nat Neurosci. 11(11):1256–8.
Walter JT, Khodakhah K. (2006) The linear computational algorithm of cerebellar Purkinje cells. J Neurosci. 13;26(50):12861–72.
Walter JT, Alviña K, Womack MD, Chevez C, Khodakhah K. (2006) Decreases in the precision of Purkinje cell pacemaking cause cerebellar dysfunction and ataxia. Nat Neurosci. 9(3):389–97.
Anita E. Autry, Ph.D.
Assistant Professor, Neuroscience
(neuroscience category)

Molecular and circuit underpinnings of social behavior
Our laboratory is focused on uncovering and dissecting neural circuits that control social behaviors and understanding how these circuits are regulated under physiological and pathological conditions. Specifically, we study parental behavior which is essential for the health and survival of offspring, as well as infant-directed aggression and other behaviors associated with parenting. The research questions center around (1) how stress affects the function of circuits controlling parental behaviors (2) how circuits that mediate stress responses interact over time and (3) how stress circuits impact feeding behavior and body composition, particularly in lactating females.
Selected Publications
Kohl, J., Babayan, B. M., Rubinstein, N.D., Autry, A. E., Marin-Rodriguez, B., Kapoor, V., Miyamaishi, K., Zweifel, L. S., Luo, L., Uchida, N., Dulac, C. (2018). Functional circuit architecture underlying parental behavior. Nature. 556 (7701) 326–331.
Kohl, J.K.*, Autry A.E.*, Dulac, C. (2017). The Neurobiology of Parenting: A Neural Circuit Perspective. Bioessays, 39(1) 1–11.
Adachi, M.*, Autry, A.E.*, Maghoub, M., Suzuki, K., Monteggia, L.M. (2017). TrkB Signaling in Dorsal Raphe Nucleus is Essential for Antidepressant Efficacy and Normal Aggression Behavior. Neuropsychopharmacology, 42(4) 886–894.
Autry, A.E. (2016). Neurobiology of Chronic Social Defeat Stress: Role of Brain-Derived Neurotrophic Factor Signaling in the Nucleus Accumbens. Biological Psychiatry, 80(6) 39–40.
Morris, M.J., Na, E.S., Autry, A.E., Monteggia, L.M. (2016). Impact of DNMT1 and DNMT3a forebrain knockout on depressive- and anxiety like behavior in mice. Neurobiology of Learning and Memory, 135; 139–145.
Renier, N., Adams, E., Kirst, C., Wu, Z., Azevedo, R., Kohl, J., Autry, A.E., Kadiri, L., Venkataraju, K.U., Zhou, Y., Wang, V.X., Tang, C.Y., Olsen, O., Dulac, C., Osten, P., Tessier-Lavigne, M. (2016). Mapping of brain activity by automated volume analysis of immediate early genes. Cell, 165 (7) 1–14.
Wu, Z., Autry, A.E., Bergan, J.F., Watabe-Uchida, M., Dulac, C.G. (2014). Galanin neurons in the medial preoptic area govern parental behavior. Nature, 509 (7500) 325–330.
Nosyreva, E., Autry, A.E., Kavalali, E.T., Monteggia, L.M. (2014). Age dependence of the rapid antidepressant and synaptic effects of acute NMDA receptor blockade. Frontiers in Molecular Neuroscience, 94; 1–7.
Nosyreva, E., Szabla, K., Autry, A.E., Ryazanov, A.G., Monteggia, L.M., Kavalali, E.T. (2013). Acute suppression of spontaneous neurotransmission drives synaptic potentiation. Journal of Neuroscience, 33(16) 6990–7002.
Benekareddy, M., Nair, A.R., Dias, B.G., Suri, D., Autry, A.E., Monteggia, L.M., Vaidya, V.A. (2013). Induction of the plasticity-associated immediate early gene Arc by stress and hallucinogens: role of brain-derived neurotrophic factor. International Journal of Neuropsychopharmacology, 16(2) 405-15.
Renata Batista-Brito, Ph.D
Assistant Professor, Neuroscience
Assistant Professor, Genetics
(neuroscience category)

Cortical development. Inhibitory circuits. Visual processing. Neurodevelopmental disorders.
Selected Publications
Batista-Brito R, Vinck M, Ferguson KA, Laubender D, Lur G, Mossner JM, Hernandez VG, Ramakrishnan C, Deisseroth K, Higley MJ, Cardin JA. Developmental dysfunction of VIP interneurons impairs cortical circuits. Neuron, 2017, 95(4):884-895.
Vinck M*, Batista-Brito R*, Knoblich U, Cardin JA. Arousal and locomotion make distinct contributions to cortical activity patterns and visual encoding. Neuron, 2015, 86(3):740-54. * equal contributions
McGinley MJ, Vinck M, Reimer MJ, Batista-Brito R, Zagha E, Cadwell CR, Tolias A S, Cardin JA, McCormick DA. Waking State: Rapid Variations Modulate Neural and Behavioral Responses. Neuron, 2015, 87(6):1143-61.
R, Rossignol E, Hjerling-Leffler J , Denaxa M, Wegner M, Lefebvre V, Pachnis V, Fishell G. The cell-intrinsic requirement of Sox6 for cortical interneuron development. Neuron, 2009, 63(4):466-81.
Tuncdemir S, Fishell G, Batista-Brito R#. miRNAs are essential for the survival and maturation of cortical interneurons. Cereb Cortex. 2014, Epub 2014 Jan 22. # corresponding author
Jaglin XH, Hjerling-Leffler J, Fishell G, Batista-Brito R#. The origin of neocortical nitric oxide synthase-expressing inhibitory neurons. Front Neural Circuits. 2012;6:44. Epub 2012 Jul 9. #corresponding author
Batista-Brito R, Machold R, Klein K and Fishell G. Gene expression in cortical interneuron precursors is prescient of their mature function. Cerebral Cortex, 2008, 18(10):2306-17.
Batista-Brito R, Fishell G. The developmental integration of cortical interneurons into a functional network. Curr Top Dev Biol, 2009, 87:81-118.
Close J, Xu H, DeMarco N, Batista-Brito R, Budy B, Fishell G. Satb1 is required for the Maturation and integration of somatostatin interneurons into the cortex. Journal of Neuroscience. 2012, 32(49):17690-705.
Picardo MA, Guigue P, Bonifazi P, Batista-Brito R, Allene C, Ribas A, Fishell G, Baude A, Cossart R. Pioneer GABA Cells Comprise a Subpopulation of Hub Neurons in the Developing Hippocampus. Neuron. 2011, 71(4):695-709.
Michael V. L. Bennett, D.Phil.
Distinguished Professor, Dominick P. Purpura Department of Neuroscience Sylvia and Robert S. Olnick Chair in Neuroscience
(neuroscience category)

Areas of investigation include: molecular and cellular physiology of glutamatergic transmission, mechanisms of delayed neurodegeneration induced by global ischemia, neuroprotection after ischemia or other insult and gap junction mediated intercellular communication.
Glutamatergic transmission is the primary mode of excitation in the nervous system. Modifications of synaptic efficacy underlie development and learning and also play important roles in disease processes. NMDA receptors, one class responding to glutamate, mediate forms of long term potentiation and depression, which can underlie memory. Protein kinases and phosphatases modify single channel properties and trafficking, i.e., movement out from the cell body, dendritic synthesis, insertion into the surface membrane, removal, and recycling or degradation. Delayed neuronal death in the hippocampal CA1 following global ischemia and in CA3 following kainate induced status epilepticus results from down regulation of GluR2, the AMPA receptor subunit that limits calcium permeability of these receptors. Increased Ca2+ influx in response to endogenous glutamate then triggers cell death by Ca2+ overload. GluR2 downregulation is mediated by REST(RE-1 silencing transcription factor), which is upregulated after ischemia. In ischemic preconditioning a brief period of ischemia leads to tolerance of a longer lasting and otherwise injurious ischemic episode. We are identifying changes in gene expression responsible for ischemic tolerance after preconditioning.
Electrical synapses formed by gap junctions synchronize many types of inhibitory interneurons in the mammalian brain. Gap junction channels are formed by a hemichannel from each of the coupled cells; because of their high conductance and permeability, it was thought that hemichannels were closed until docking with another hemichannel. Now it there is evidence that hemichannels not apposed to another hemichannel can open under physiological as well as pathological conditions. We are investigating the controlling mechanisms at the level of single (hemi) channels. Hemichannels mediate intercellular signaling by secreted molecules, such as ATP, and may be involved in propagation of damage (or protection) at boundaries between normal and injured tissue. Several human diseases are caused by connexin mutations, including X-linked Charcot-Marie-Tooth disease, one type of non-syndromic deafness, one type of epilepsy, two types of cataract, and oculodentodigital dysplasia (ODDD). We are analyzing how the altered biophysics of the mutations leads to the pathology.
Selected Publications
Eugenin EA, King JE, Hazleton JE, Major EO, Bennett MVL, Zukin RS, Berman JW. (2011) Differences in NMDA receptor expression during human development determine the response of neurons to HIV-Tat-mediated neurotoxicity. Neurotox. Res. 19: 138–148. PMID: 20094923.
Orellana JA, Froger N, Ezan P, Jiang JX, Bennett MVL, Naus CC, Giaume C, Sáez JC. (2011) ATP and glutamate released via astroglial connexin43 hemichannels mediate neuronal death through activation of pannexin 1 hemichannels. J Neurochem. Feb 5. doi: 10.1111/j.1471–4159.2011.07210.x. [Epub ahead of print] PMID: 21294731.
Stetler, R.A., Gao, Y., Zukin, R.S., Vosler, P.S., Zhang, L., Zhang, F., Cao, G., Bennett, M.V.L., Chen, J. (2010) Apurinic/apyrimidinic endonuclease APE1 is required for PACAP-induced neuroprotection against global cerebral ischemia. Proc. Natl. Acad. Sci. U.S.A. 107: 3204–3209. PMID: 20133634.
Palacios-Prado, N., Biggs, S.W., Skeberdis, V.A., Pranevicius, M., Bennett, M.V.L., Bukauskas, F.F. (2010) pH-dependent modulation of voltage gating in connexin45 homotypic and connexin45/connexin43 heterotypic gap junctions. Proc. Nat. Acad. Sci. U.S.A. 107: 9897–9902. PMID: 20445098.
Sáez JC, Schalper KA, Retamal MA, Orellana JA, Shoji KF, Bennett MVL. (2010) Cell membrane permeabilization via connexin hemichannels in living and dying cells. Exp Cell Res. 316: 2377–89. PMID: 20565004.
Garré JM, Retamal MA, Cassina MP, Barbeito L, Bukauskas FF, Sáez JC, Bennett MVL, Abudara V. (2010) FGF-1 induces ATP release from spinal astrocytes in culture and opens pannexin and connexin hemichannels. Proc. Natl. Acad. Sci. U.S.A. 107: 22659–22664. PMID: 21148774.
Pablo E. Castillo, M.D., Ph.D.
Professor, Dominick P. Purpura Department of Neuroscience Harold and Muriel Block Chair in Neuroscience
(neuroscience category)

Synaptic transmission underlies every aspect of nervous system function. How we think, feel, act and learn, all rely on information transfer between nerve cells. In addition, synapses are extremely dynamic, and activity-dependent changes in synaptic strength are essential to most forms of learning. It is becoming increasingly clear that synaptic dysfunction is central to the etiology and progression of a wide range of neuropsychiatric and neurodevelopmental disorders. The main goal of my research program is to understand the cellular and molecular basis of activity-dependent changes in synaptic strength at both excitatory and inhibitory connections, and how such changes are modified during pathological conditions. In our studies we use brain slice electrophysiology and pharmacology, two-photon laser microscopy, optogenetics and a wide-range of molecular manipulations. To gain insights into the mechanisms of synaptic function, we include in our studies functional analyses of transgenic mice for several synaptic proteins, as well as mouse models for various neuropsychiatric conditions, including Alzheimer’s disease, autistic spectrum disorders and schizophrenia.
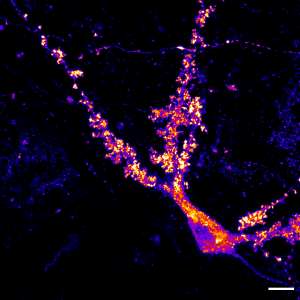
Mossy cell in the dentate gyrus of the hippocampus expressing labelled actin in its large postsynaptic structures, the thorny excresences.
Selected Publications
Hashimotodani Y, Nasrallah K, Jensen KR, Chávez AE, Carrera D, Castillo PE. (2017) LTP at Hilar Mossy Cell-Dentate Granule Cell Synapses Modulates Dentate Gyrus Output by Increasing Excitation/Inhibition Balance. Neuron 95:928-9.
Monday HR and Castillo PE (2017) Closing the gap: long-term presynaptic plasticity in brain function and disease. Curr Opin Neurobiol 45:106-112.
Younts TJ, Monday HR, Dudok B, Klein ME, Jordan BA, Katona I, Castillo PE. (2016) Presynaptic Protein Synthesis Is Required for Long-Term Plasticity of GABA Release. Neuron 92:479-492.
Park J, Chávez AE, Mineur YS, Morimoto-Tomita M, Lutzu S, Kim KS, Picciotto MR, Castillo PE, Tomita S. (2016) CaMKII Phosphorylation of TARPγ-8 Is a Mediator of LTP and Learning and Memory. Neuron. 2016 Oct 5;92(1):75-83.
Jurgensen S, Castillo PE. (2015) Selective Dysregulation of Hippocampal Inhibition in the Mouse Lacking Autism Candidate Gene CNTNAP2. J Neurosci 35:14681-7.
Klein ME, Castillo PE, Jordan BA (2015) Coordination between translation and degradation regulates inducibility of mGluR-LTD. Cell Reports 10:1459–66.
Younts TJ, Castillo PE (2014) Endogenous cannabinoid signaling at inhibitory interneurons. Curr Opin Neurobiol. 26:42-50.
Klein ME, Younts TJ, Castillo, PE, Jordan, BA (2013) RNA-binding protein Sam68 controls synapse number and local β-actin mRNA metabolism in dendrites. Proc Nat Acad Sci USA. 110(8):3125-3130.
Kaeser-Woo YJ, Younts TJ, Yang X, Zhou P, Wu D, Castillo PE, Südhof TC (2013) Synaptotagmin-12 phosphorylation by cAMP-dependent protein kinase is essential for hippocampal mossy fiber LTP. J. Neurosci. 33(23):9769-9780.
Younts TJ, Chevaleyre V, Castillo PE (2013) CA1 pyramidal cell theta-burst firing triggers endocannabinoid-mediated long-termdepression at both somatic and dendritic inhibitory synapses. J. Neurosci. 33:13743-13757.
Kostantin Dobrenis, Ph.D.
Assistant Professor, Dominick P. Purpura Department of Neuroscience
(neuroscience category)

Our principal interests lie in the pathogenesis and therapy of neurodegenerative diseases, and in the fields of ganglioside and microglial biology. We have contributed to the characterization of animal models of neuronal lysosomal storage diseases including Tay-Sachs/Sandhoff disease, Niemann Pick C (NPC) disease, mucopolysaccharidosis (MPS) IIIA, and mucolipidosis IV, and more recently to understanding the pathogenesis of Christianson Syndrome, a sodium/hydrogen exchanger deficiency that displays features of storage disorders. Much of our work has been directed at developing rational therapeutic strategies for these and related genetic diseases which affect the central nervous system (CNS) in a global manner. The goal here is to find ways to effectively replace the missing protein, or compensate for its function, within cells throughout the CNS. This entails overcoming challenges such as the blood brain barrier, and developing strategies that enhance neuronal uptake of therapeutic compounds.
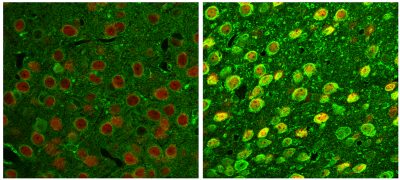
Immunofluorescent staining of neocortex from wild type (left) and Niemann Pick C disease mice showing significant upregulation and altered distribution of an ion channel (green) in neurons (NeuN-positive; red) not previously studied in this disease, but implicated by gene expression studies by the lab also suggesting its correction by cyclodextrin therapy of disease mice.
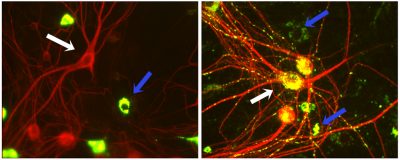
Neuronal cultures incubated with a soluble green reporter protein as is (left), or coupled to a peptide (right), developed by the lab, to enhance neuronal endocytic uptake and delivery to lysosomes for enzyme replacement therapy for lysosomal storage diseases. Uptake of the fluorescent protein (green) without the peptide (left) by neurons (red; positive for MAP2-immunostaining; e.g. white arrows) is virtually undetectable in contrast to more endocytically active glia (MAP2-negative; blue arrows). With coupled peptide (right), neuronal uptake is dramatically enhanced.
One of our ongoing projects in this regard is the development of fusion genes of hexosaminidase, the enzyme deficient in Tay Sachs disease, and peptide sequences related to the atoxic fragment of tetanus toxin. Due to characteristics of the latter, the encoded chimeric proteins have properties allowing circumvention of the blood brain barrier, increased neuronal endocytotic uptake into the lysosomal compartment, and transneuronal transsynaptic trafficking for wider dissemination of the needed protein. Additional projects include: exploring the efficacy and delineating the mechanisms of action of small molecule therapies such as miglustat, related analogs, and cyclodextrin for storage diseases, now seeing human application, in cellular, biochemical, gene expression and behavioral assays; and investigation of novel contact-mediated mechanisms of neuronal-microglial lysosomal enzyme transfer for effective hematopoietic stem cell replacement CNS therapy. Furthermore we continue to be engaged in studies examining the role and expression patterns of gangliosides and microglia in development and neuropathology. The lab utilizes techniques ranging from molecular recombinant work to animal behavioral assays with extensive experience in: cell culture preparations of all the major CNS cell types; gangliosides and lysosomal enzyme biochemistry; vital and fixed specimen histologic and immunocytochemical techniques; modern fluorescent techniques for monitoring organellar or biochemical activities in living cells; and a wide range of high resolution imaging and image analysis techniques.
Selected Publications
Boudewyn, L.C., Sikora, J., Kuchar, L., Ledvinova, J., Grishchuk, Y., Wang, S., Dobrenis, K., Walkley, S.U. N-butyl-deoxynojirimycin delays motor deficits, cerebellar microgliosis and Purkinje cell loss in a mouse model of mucolipidosis type IV. Neurobiol. Disease, 105:257-270, 2017.
Trilck, M., Peter, F., Zheng, C., Frank, M., Dobrenis, K., Mascher, H., Rolfs, A., Frech, M.J. Diversity of glycosphingolipid GM2 and cholesterol accumulation in NPC1 patient-specific iPSC-derived neurons. Brain Res. 1657:52-61, 2017.
Yang, D.-S., Stavrides, P., Kumar, K., Jiang, Y, Mohan, PS., Ohno, M., Dobrenis, K., Davidson, C.D., Saito, M, Pawlik, M., Huo, C., Walkley, S.U., Nixon, R.A. Cyclodextrin has conflicting actions on autophagy flux in vivo in brains of normal and Alzheimer model mice. Human Mole. Genet. 26:843-859, 2017.
Davidson, C., Fishman, YI, Puskas I., Szeman, J., Sohajda, T., McCauliff, L.A., Sikora, J., Storch, J., Vanier M.T., Szente, L., Walkley, S.U., Dobrenis, K. Efficacy and ototoxicity of different cyclodextrins in Niemann-Pick C disease. Ann Clin Transl Neurol. 3:366-380, 2016.
Saito, M., Wu, G., Hui, M., Masiello, K., Dobrenis, K., Ledeen, R.W., Saito, M. Ganglioside accumulation in activated glia in the developing brain: comparison between WT and GalNacT KO mice. J. Lipid Res. 56:1434-1448, 2015.
Farfel-Becker, T., Vitner, E.B., Kelly, S.L., Bame, J.R., Duan, J., Shinder, V., Merrill Jr, A.F., Dobrenis, K., Futerman, A.H. Neuronal accumulation of glucosylceramide in a mouse model of neuronopathic Gaucher disease leads to neurodegeneration. Human Molec. Genet 23:843-854, 2014.
Micsenyi, M.C., Sikora, J., Stephney, G., Dobrenis, K., Walkley, S.U. Lysosomal membrane permeability stimulates protein aggregate formation in neurons of a lysosomal disease. J. Neurosci. 33:10815-10827, 2013.
Erblich, B., Zhu, L., Etgen, A., Dobrenis, K., Pollard, J.W. Absence of colony stimulating factor-1 receptor signaling results in loss of microglia, disrupted brain development and olfactory deficits. PloS One, 2011. 2011;6(10):e26317. Epub 2011 Oct 27.
Stromme, P., Dobrenis, K., Sillitoe, R.V., Gulinello, M., Ali, N.F., Davidson, C., Micsenyi, M.C., Stephney, G., Ellevog, L., Klunglund, A., Walkley, S.U. X-linked Angelman-like syndrome caused by Slc9a6 knockout in mice exhibits evidence of endosomal-lysosomal dysfunction. Brain 134:3369-83, 2011.
Gulinello, M., Chen, F., Dobrenis, K. Early deficits in motor coordination, cognitive function and anxiety-like behavior in a mouse model of the neurodegenerative lysosomal storage disorder, Sandhoff disease. Behav. Brain Res. 193:315-319, 2008.
Anna Francesconi, Ph.D.
Associate Professor, Dominick P. Purpura Department of Neuroscience
(neuroscience category)

Molecular mechanisms of metabotropic glutamate receptor function.
Research in the laboratory focuses on elucidating the molecular and cellular underpinnings of metabotropic glutamate receptor function in the brain, with the ultimate goal of developing a molecular rationale for targeted interventions in neuropsychiatric disorders. A growing body of evidence from studies in human subjects and animal models has established a link between dysfunctions in glutamatergic neurotransmission and developmental brain abnormalities associated with intellectual disability, autism and schizophrenia. Group I metabotropic glutamate receptors, mGlu1 and mGlu5, are G protein-coupled receptors critical to the formation and maintenance of brain circuitry and activity-dependent synaptic plasticity, a cellular substrate of learning and memory. Dysregulation of group I mGlu receptor activity is implicated in neurodevelopmental disorders including Fragile X syndrome and schizophrenia.
We use a combination of molecular biology, biochemistry and imaging techniques to uncover the molecular mechanisms underlying temporo-spatial regulation of mGluR signaling and to examine mGluR functions in neuronal homeostasis and synaptic plasticity. Ongoing studies pursue interrelated lines of investigation by examining the role of adaptor proteins in orchestrating and fine-tuning mGluR activity under physiological conditions and in animal models of Fragile X syndrome; and by investigating the cellular mechanisms by which mGluR signaling contributes to synaptogenesis and neuronal maturation.
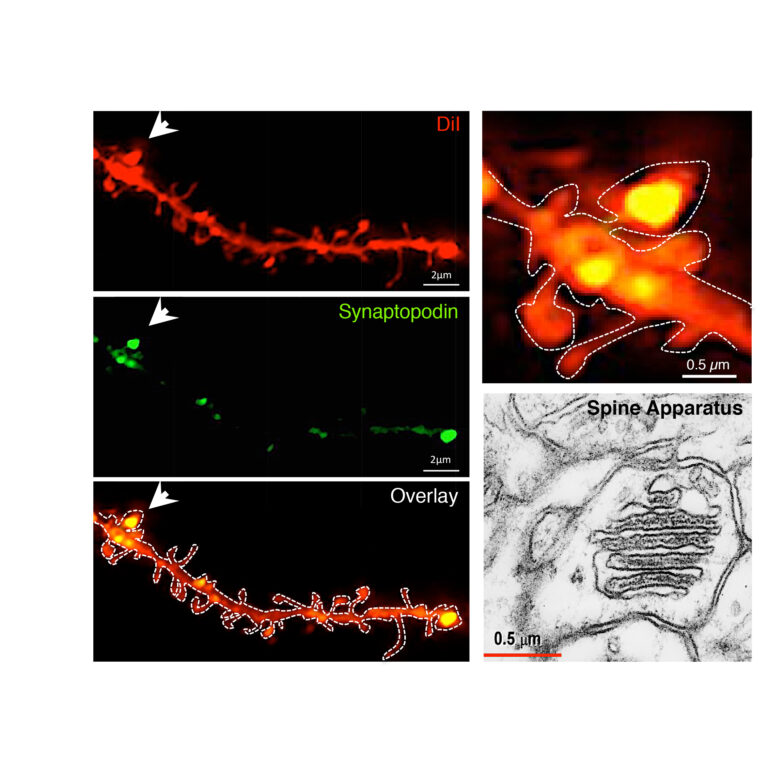
Group I mGluRs are enriched in spines endowed with Spine Apparatus. A subset of telencephalic mushroom-type spines is endowed with a spine apparatus, an organelle composed of sER stacks intercalated by electron-dense plates formed by synaptopodin and actin. Visualization of synaptopodin in dendritic spines of hippocampal neurons labeled with DiI: confocal images illustrate synaptopodin presence within the head of mature mushroom spines. Ultrastructure of the spines apparatus visualized by electron microscopy (from the SynapseWeb).
Selected Publications
Mende M, Fletcher EV, Belluardo JL, Pierce JP, Bommareddy PK, Weinrich JA, Kabir ZD, Schierberl KC, Pagiazitis JG, Mendelsohn AI, Francesconi A, Edwards RH, Milner TA, Rajadhyaksha AM, van Roessel PJ, Mentis GZ, Kaltschmidt JA. (2016) Sensory-Derived Glutamate Regulates Presynaptic Inhibitory Terminals in Mouse Spinal Cord. Neuron 90: 1189-1202. PMID: 27263971.
Kalinowska, M., and Francesconi A. (2016) Group I Metabotropic Glutamate Receptor Interacting Proteins: Fine-Tuning Receptor Functions in Health and Disease. Current Neuropharmacology 14(5): 494-503. PMID: 27296642.
Kalinowska, M., Chavez, A.E., Lutzu, S., Castillo, P.E., Bukauskas, F.F., and Francesconi. A. (2015) Actinin-4 Governs Dendritic Spine Dynamics and Promotes their Remodeling by Metabotropic Glutamate Receptors. J. Biol. Chem. 290: 15909-20. PMCID: PMC4481196.
Kalinowska, M., Castillo, C., and Francesconi A. (2015) Quantitative profiling of brain lipid raft proteome in a mouse model of fragile x syndrome. PLoS One. Apr 7;10(4):e0121464. PMCID: PMC4388542
Kumari, R., Castillo, C., and Francesconi, A. (2013) Agonist-dependent signaling by group I metabotropic glutamate receptors is regulated by association with lipid domains J. Biol. Chem. 288: 32004-32019. PMCID: PMC3814796
Kumari, R., and A. Francesconi. (2011). Identification of GPCR localization in Detergent-Resistant Membranes. Methods Mol. Biol. 746:411-423.
Takayasu, Y., Takeuchi, K., Kumari, R., Bennett, M.V., Zukin, R.S., and A. Francesconi. (2010) Caveolin-1 knockout mice exhibit impaired induction of mGluR-dependent long-term depression at CA3-CA1 synapses. Proc. Natl. Acad. Sci. U S A. 107:21778-83.
Francesconi, A., Kumari, R. and R.S. Zukin. (2009). Proteomic analysis reveals novel binding partners of metabotropic glutamatereceptor 1. J. Neurochem. 108:1515-1525.
Francesconi, A., Kumari, R. and R.S. Zukin. (2009). Regulation of group I mGluR trafficking and signaling by the caveolar/lipid raft pathway. J. Neurosci. 29:3590-3602.
Castillo PE, Francesconi A, Carroll RC. (2008) The ups and downs of translation-dependent plasticity. Neuron 59: 1-3. PMID: 18614022.
Tiago Gonçalves, Ph.D.
Assistant Professor, Dominick P. Purpura Department of Neuroscience
(neuroscience category)

The dentate gyrus (DG) is the main input region of the hippocampus. As such, it plays a crucial role in hippocampal function, including learning and memory. Additionally, the DG is one of only two regions of the mammalian brain that continuously add new neurons through adulthood, and there is increasing evidence that these adult-born neurons play a role in specific learning tasks: particularly, the ability to distinguish between similar memories. Adult neurogenesis constitutes an unusual mode of plasticity in the brain, with specific stimuli such as environmental enrichment and voluntary exercise modulating the number of neurons integrating DG networks. In addition, adult-born neurons have been used to study neuronal development, and defects in neurogenesis have been associated with several human neurological and psychiatric diseases. The main focus of my research interests is to understand how enrichment and activity regulate the proliferation of adult neural stem cells and shape nascent adult-born neurons during their integration into DG circuits.
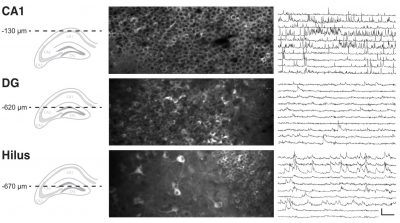
In vivo Ca2+ imaging of different hippocampal areas 3-photon images and Ca2+ traces of neurons expressing a genetically encoded Ca2+ sensor in CA1, DG granule layer and hilus/sub-granular layer. Note differences in morphology and activity patterns betwen cells in these layers. Scale bars: 100% ∆F/F 20 s.
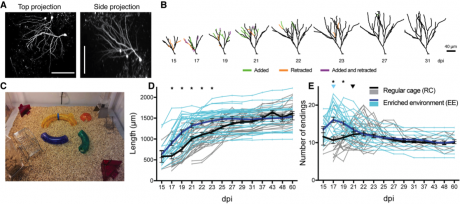
Exposure to an enriched enviroment (EE) leads to faster dendritic growth and earlier pruning (A) In vivo 2-photon images of RV-GFP labeled cells in the DG imaged 60 days post-infection (dpi). Scale bar = 100 µm. (B) Representative reconstructions of the dendrites of two newborn DGCs, branches added and retracted between imaging time points are highlighted. (C) EE cage is large (91×91) cm) and contains running wheels. (D) Individual and mean (bold) plot of dendrite length. (D) Branching of EE dendrites peaks earlier and maximum number of endings is higher than in RC dendrites (adapted from Gonçalves et al. 2016)
Experience and the activity it elicits are powerful modulators of neuronal development. As the brain matures it undergoes several critical periods where specific behavioral experiences (for example environmental enrichment and exercise) are needed for correct circuit formation. In the adult brain, behavioral experience has been shown to influence the proliferation of neural stem cells in the subgranular layer of the DG, their differentiation into glia and neurons, the maturation of these adult-born neurons, and their connectivity and incorporation into hippocampal circuits. Yet many aspects of the effects of experience on adult neurogenesis and hippocampus networks remain poorly understood. In our lab we study the effects of activity and experience at different phases of neuronal development ranging from the neural precursor stage to full maturity. The ability to track individual cells over their maturation period is crucial for our experiments. We use in vivo 2-photon imaging as a powerful tool to study of neuronal development since it allows for the recurrent probing of individual cells and circuits while keeping sensory input, neuronal activity patterns, and the physiological environment intact.
Selected Publications
Mansour A.F., Gonçalves J.T., Bloyd C.W., Li H., Fernandes S., Quang D., Johnston S., Parylak S.L., Jin X, Gage F.H., An in vivo model of functional and vascularized human brain organoids, Nat. Biotechnol. , doi:10.1038/nbt.4127 (2018)
O’Donnell C., Gonçalves J.T., Portera-Cailliau C., Sejnowski T.J., Beyond excitation/inhibition imbalance in multidimensional models of neural circuit changes in brain disorders, eLife, 6:e26724 (2017)
O’Donnell C., Gonçalves J.T., Whiteley N., Portera-Cailliau C., Sejnowski T.J., The population tracking model: A simple, scalable statistical model for neural population data, Neural Computation, 29, 50–93 (2017)
Gonçalves J. T. *, Schafer S.T. * (equal contribution), Gage F.H., Adult neurogenesis in the hippocampus: from stem cells to behavior, Cell, 167, 897–914 (2016) review article
Gonçalves J.T., Bloyd C.W., Shtrahman M, Johnston S.T., Schafer S.T., Parylak S.L., Thanh T., Chang T., Gage F.H., In vivo imaging of dendritic pruning in dentate granule cells, Nat. Neurosci., 19(6), 788–791 (2016) (journal cover)
Poo M.M., Pignatelli M., Ryan T.J., Tonegawa S., Bonhoeffer T., Martin K.C., Rudenko A., Tsai L., Tsien R.W., Fishell G., Mullins C., Gonçalves J.T., Shtrahman M., Johnston S.T., Gage F.H., Dan Y., Long J. Buzsáki G., Stevens C., What is memory? The present state of the engram, BMC Biology, 14(1):40 (2016) opinion article
Mertens J., Paquola A. C. M., Ku M., Hatch E., Böhnke L., Ladjevardi S., McGrath S., Campbell B., Lee H., Herdy J. R., Gonçalves J. T., Toda T., Kim Y., Winkler J., Yao J., Hetzer M. and Gage F. H., Directly reprogrammed human neurons retain age-related transcriptomic signatures and reveal age-related nucleocytoplasmic defects, Cell Stem Cell, 17, 1–14 (2015)
Gonçalves J.T., Anstey J.E., Golshani P., Portera-Cailliau C., Circuit level defects in the developing neocortex of fragile X mice, Nat. Neurosci., 16, 903–09 (2013)
(equal contribution) Cheng A.*, Gonçalves J.T. *, Golshani P., Arisaka K., Portera-Cailliau C., Spatio-temporal excitation-emission multiplexing for multiple beam 2-photon calcium imaging in deep tissue, Nat. Methods., 8, 139–42 (2011)
Golshani P., Gonçalves J.T., Khoshkhoo S., Mostany R., Smirnakis S., Portera-Cailliau C., Internally mediated developmental desynchronization of neocortical network activity, J. Neurosci., 29, 10890–10899 (2009)
David H. Hall, Ph.D.
Professor, Dominick P. Purpura Department of Neuroscience
(neuroscience category)

The soil nematode Caenorhabdidis elegans is a model system used to study the genetic control of cellular development. The Hall laboratory specializes in ultrastructural studies of the C. elegans. We use serial thin sections, electron microscopy, electron tomography, FIB/SEM and immunocytochemistry as primary tools to follow the development of identified neurons, particularly their axon outgrowth and synaptic connectivity. We also conduct collaborative studies on many other tissues in the embryo, larval, dauer, adult and aging nematode, including many epithelial tissues and the germline.
We host the Center for C. elegans Anatomy, supported by the NIH Office of the Director, and train students in anatomical methods for this system. Members of the lab are authoring the website www.WormAtlas.org. It displays nematode anatomy in great detail through multiple applications including Slidable Worm, a Handbook of all cells and tissues, the WormImage catalogue, a Glossary, and selected html texts of classic papers.
In collaboration with Scott Emmons, we are studying the complete connectome of C. elegans in both sexes and in larval stages to uncover how the nematode wiring diagram develops over time. In collaboration with Maureen Barr (Rutgers) we are studying the “tubulin code” which helps to stabilize ciliary microtubules during development and maintenance of the nematode’s sense endings.
Selected Publications
Whole-animal connectomes for both Caenorhabditis elegans sexes (2019) Cook, S.J., Jarrell, T.A., Brittin, C., Wang, Y., Bloniarz, A.E., Yakovlev, M.A., Nguyen, K.C.Q., Tang, L.T-H., Bayer, E.A., Duerr, J.S., Bülow, H.E., Hobert, O., Hall, D.H. & Emmons, S.W. Nature 571: 63–71.
Axon-Dependent Patterning and Maintenance of Somatosensory Dendritic Arbors (2019). Ramirez-Suarez NJ, Belalcazar HM, Salazar CJ, Beyaz B, Raja B, Nguyen KCQ, Celestrin K, Fredens J, Færgeman NJ, Hall DH, Bülow HE. Dev Cell. 48:229–244.
The AFF-1 exoplasmic fusogen is required for endocytic scission and seamless tube elongation (2018). Soulavie F, Hall DH, Sundaram MV. Nat Commun. 9(1):1741. doi: 10.1038/s41467-018-04091-1.
C. elegans neurons jettison protein aggregates and mitochondria under neurotoxic stress (2017) Melentijevic I, Toth ML, Arnold ML, Guasp RJ, Harinath G, Nguyen KC, Taub D, Parker JA, Neri C, Gabel CV, Hall DH, Driscoll M. Nature. 542: 367–371.
Cell-specific α-tubulin isotype regulates ciliary microtubule ultrastructure, intraflagellar transport, and extracellular vesicle biology (2017). Silva M., Morsci N., Nguyen K.C., Rizvi A., Rongo C, Hall D.H., Barr M.M.Curr Biol. pii: S0960-9822(17)30208-7
Glia-derived neurons are required for sex-specific learning in C. elegans (2015) Sammut, M., Cook, S.J., Nguyen, K., Felton, T., Hall, D.H., Emmons, S.W., Poole, R.J. and Barrios, A. Nature 526: 385–90.
Neurite sprouting and synapse deterioration in the aging C. elegans nervous system (2012) Toth, M., Melentijevic, I., Shah, L., Bhatia, A., Lu, K., Talwar, A., Naji, H., Ibanez-Ventoso, C., Ghose, P., Jevince, A., Xue, J., Herndon, L.A., Bhanot, G., Rongo, C., Hall, D.H., and Driscoll, M. J. Neurosci. 32: 8778-90.
The connectome of a decision making neuronal network (2012) Jarrell, T.A., Wang, Y., Bloniarz, A.E., Brittin, C.A., Xu, M., Thomson, J.N., Albertson, D.G., Hall, D.H. and Emmons, S.W. Science 337: 437– 444.
Stochastic and genetic factors influence tissue-specific decline in ageing C. elegans (2012) Herndon, L.A., Schmeissner, P.J., Dudaroneck, J.M., Brown, P.A., Listner, K.M., Paupard, M.C., Hall, D.H., and Driscoll, M. Nature 419: 788–794.
Jean M. Hébert, Ph.D.
Professor, Dominick P. Purpura Department of Neuroscience Professor, Department of Genetics
(neuroscience category)

Generating and regenerating the neocortex
The Hébert lab has traditionally studied how the forebrain develops using conditional genetic methods in mice. Recently, the focus of the lab has transitioned to two new areas of interest. First, we are studying how homeostasis is maintained in the adult forebrain using primarily molecular genetic techniques to manipulate the expression of regulatory genes in neural stem and progenitor cells. Specifically, we are examining how neurogenesis is maintained in the hippocampus, how the myelination of axons is maintained through continuous oligodendrogenesis, and how cells become reactive to brain damage.
Second, we are devising novel methods to regenerate the damaged neocortex, the part of our brains that we use for our highest cognitive and perceptual functions. Neocortical damage can be local, due for example to stroke or trauma, or widespread, due for example to neurodegeneration or aging. Among the approaches we are taking, we are developing ways of replacing the principle neurons of the adult neocortex without significantly disrupting the function of existing neural circuits. These approches involve the use of mixed cell populations for transplantation and for widespread damage the use of cells that can disperse throughout the neocortex to repopulate and bolster existing neural circuits with new cells.
Selected Publications
Kamatkar N, Levy M, Hébert JM. (2019). Development of a monomeric inhibitory RNA aptamer specific for FGFR3 that acts as an activator when dimerized. Mol. Ther. Nuc. Acids, in press.
Kang W. Nguyen KCQ, Hébert JM. (2019). Transient redirection of SVZ-stem cells to oligodendrogenesis by FGFR activation promotes remyelination. Stem Cell Reports 12:1223-1231.
A recent blog: http://blogs.einstein.yu.edu/the-science-of-replacement-as-a-means-of-escaping-aging/
Hébert JM, Vijg J. (2018). Cell replacement to reverse brain aging: challenges, pitfalls, and opportunities. Trends in Neuroscience 41: 267-279.
Antoine MW, Zhu X, Dieterich M, Brandt T, Vijayakumar S, McKeehan N, Arezzo J, Zukin RS, Borkholder D, Jones SM, Frisina R, Hébert JM. (2018). Left-right brain lateralization in mammals due to early uneven ear input. PLoS Biology 16: e2002988.
Nandi S, Alvina K, Lituma PJ, Castillo PE, Hébert JM. (2018). Neurotrophin and FGF signaling adapter proteins, FRS2 and FRS3 regualte dentate granule cell maturation and exitatory synaptogenesis. Neuroscience 369: 192:201.
Nandi S, Gutin G, Blackwood CA, Kamatkar NG, Lee KW, Fishell G, Wang F, Goldfarb M, Hébert JM. (2017). Context-driven, receptor-dependent usage of an intracellular adapter governs specificity in FGF signal transduction. J. Neurosci. 37:5690-5698.
Antoine MW, Vijayakumar S, McKeehan N, Jones S, Hébert, JM. (2017). The severity of vestibular dysfunction in deafness as a determinant of comorbid hyperactivity or anxiety. J. Neurosci. 37: 5144-5154.
Andriani GA, Faggioli F, Baker D, Dollé MET, Sellers RS, Hébert JM, van Steeg H, Hoeijmakers J, Vijg J, Montagna C. (2016). Whole chromosome aneuploidy in the brain of BubR1H/H and Ercc1 -/Δ7 mice. Hum. Mol. Gen. 25: 755-765.
Nandi S, Chandramohan D, Fioriti L, Melnick AM, Hébert JM, Mason CE, Kandel ER, Rajasethupathy P. (2016). A role for piRNAs in retrotransposon silencing in the mammalian brain. P.N.A.S. U.S.A. 113: 12696-12702.
Kang W, Hébert JM. (2015). FGF signaling is necessary for neurogenesis in young mice and sufficient to reverse its decline in old mice. J. Neuroscience 35: 10217-10223.
Bryen A. Jordan, Ph.D.
Associate Professor, Dominick P. Purpura Department of Neuroscience Associate Professor, Psychiatry and Behavioral Sciences
(neuroscience category)

Exploring synaptic function and activity-dependent synapse-to-nucleus signaling
An important question in neuroscience is how does neuronal activity alter neuronal connectivity. This question is critically important since changes in connectivity and transmission underlie higher order brain functions such as learning and memory and likely play a role in the cognitive deficits observed in many neurological diseases. To explore this question, we employ proteomics and mass spectrometry, which provide us with a global overview of synaptic and nuclear complexity and allow us to explore their dynamics. Using these methods, we found that a number of synaptic components can shuttle to the neuronal nucleus in response to synaptic activity. These include PRR7 and AIDA-1, which binds to NMDA receptors (NMDAR) and links synaptic activity to nuclear functions. Recent studies implicate AIDA-1 in diverse psychiatric and developmental disorders including schizophrenia and Autism spectrum disorders. A single nucleotide polymorphism (SNP) in the AIDA-1 gene (ANKS1b) is associated with response to antipsychotics, suggesting AIDA-1 may play a role in schizophrenia. Moreover copy number variations (CNVs) and SNPs of AIDA-1 have been identified in patients with autism and correlate positively with impaired play skills in ASD. Moreover we have recently found that AIDA-1 can regulate the metabolism of the Amyloid Precursor Protein (APP) in neurons. AIDA-1 can promote the generation of amyloid beta peptides by regulating APP internalization, and may therefore it may play an important role in Alzheimer’s disease.
Moreover we found that certain RNA binding proteins (RNABPs) shuttle back into synaptic junctions in response to neuronal activity. We have recently shown that one of these proteins, Sam68, regulates the synaptic and dendritic expression of beta-actin and is crucial for proper spine morphology and synaptic function. Sam68 has been recently implicated in Fragile X-associated Tremor/Ataxia Syndrome (FXTAS), which is a neurodegenerative disorder caused by mutations upstream of the FMR1 gene. We are therefore investigating if Sam68-dependent protein translation of cytoskeletal components can affect synaptic function and plasticity and ultimately behavior. We believe Sam68 plays a role in the generation and refining of neuronal networks. Understanding precisely how neurons regulate specific connections amongst their many thousand inputs is a central question in neuroscience. Therefore our lab employs broad-based proteomics methods to understand how synapses relay fast synaptic information to the nucleus and back, what are the key players in this process, and what role do these molecules play in brain pathologies.
Selected Publications
Younts TJ, Monday HR, Dudok B, Klein ME, Jordan BA, Katona I, Castillo PE. Presynaptic protein synthesis is required for long-term plasticity of GABA release. Neuron 2016 Oct 19; 92(2):479–492
Kravchick DO, Karpova A, Hrdinka M, Lopez-Rojas J, Iacobas S, Carbonell AU, Iacobas DA, Kreutz MR, Jordan BA. Synaptonuclear messenger PRR7 inhibits c-Jun ubiquitination and regulates NMDA mediated excitotoxicity. The EMBO Journal. 2016 Sep 1;35(17):1923–34
Tindi JO, Chávez AE, Cvejic S, Calvo-Ochoa E, Castillo PE, Jordan BA. ANKS1B Gene Product AIDA-1 Controls Hippocampal Synaptic Transmission by Regulating GluN2B Subunit Localization. J. Neurosci.2015 Jun 17;35 (24), 8986–8996
Kravchick DO, Jordan BA. Presynapses go nuclear! EMBO J. 2015 Apr 15;34(8):984–6.
KleinME, Castillo PE, Jordan BA. Coordination between Translation and Degradation Regulates Inducibility of mGluR-LTD. Cell Reports 2015 Mar 10;(10):1459–1466
Klein ME, Younts TJ, Castillo PE, Jordan BA. RNA-binding protein Sam68 controls synapse number and local β-actin mRNA metabolism in dendrites. PNAS 2013 Feb 19;110(8):3125–30
Mulholland PJ, Jordan BA, Chandler LJ. Chronic ethanol up-regulates the synaptic expression of the nuclear translational regulatory protein AIDA-1 in primary hippocampal neurons. Alcohol. 2012 Sep;46(6):569–76.
Zhang G, Neubert TA, Jordan BA. RNA binding proteins accumulate at the postsynaptic density with synaptic activity. J Neurosci. 2012 Jan 11:32(2): 599–609.
Jacob AL, Jordan BA, Weinberg RJ. Organization of amyloid-beta protein precursor intracellular domain-associated protein-1 in the rat brain. J Comp Neurol. 2010 Aug 15;518(16):3221–36.
Jordan BA, Kreutz MR. Nucleocytoplasmic protein shuttling: the direct route in synapse-to-nucleus signaling. Trends in Neurosci (TINS). 2009 Jul;32(7):392–401.
Adam Kohn, Ph.D.
Professor, Dominick P. Purpura Department of Neuroscience Professor, Department of Ophthalmology and Visual Sciences Professor, Departmen of Systems and Computational Biology Isidor Tachna Professor in Ophthalmology
(neuroscience category)

Our laboratory studies the neural circuits that underlie visual perception, a general issue that we approach from several directions. For instance, we study how the responsivity and tuning of cortical neurons is altered by recent stimulus history. This form of rapid plasticity—termed adaptation—has strong perceptual effects, allowing us to explore the neurophysiological underpinnings of perceptual phenomena. In addition, we are interested in understanding the functional benefit of adaptation and in learning how adaptation early in the visual system affects subsequent stages of processing. We hope that by understanding the principles of adaptation we will also gain insight into other forms of plasticity such as perceptual learning and recovery from injury. We also study how populations of neurons function together to encode information about the visual world. We record from small populations of neurons simultaneously and measure the correlation of their responses. In particular, we explore how correlation depends on stimulus parameters, recent stimulus history, and cortical location. The primary techniques of the lab are neurophysiological recordings, computational modeling, and psychophysics. We hope that employing a range of experimental techniques will help us understand the computations carried out by the visual system and the circuits that perform them.
Selected Publications
Coen-Cagli R, Kohn A*, Schwartz O* (2015) Flexible gating of contextual influences in natural vision. Nature Neuroscience 18: 1648–1655. *Equal contribution
Zandvakili A, Kohn A (2015) Coordinated neuronal activity enhances corticocortical communication. Neuron 87: 827–839.
Solomon SG, Kohn A (2014) Moving sensory adaptation beyond suppressive effects in single neurons. Current Biology 24: 1012–1022.
Czuba TB, Huk AC, Cormack LK, Kohn A (2014) Area MT encodes three-dimensional motion. Journal of Neuroscience 34: 15522–15533.
Patterson CA, Wissig SC, Kohn A (2014) Adaptation disrupts motion integration in the primate dorsal stream. Neuron 81: 674–686.
Jia X, Tanabe S, Kohn A (2013) Gamma and the coordination of spiking activity in early visual cortex. Neuron 77: 762–774.
Smith MA, Jia X, Zandvakili A, Kohn A (2013) Laminar dependence of neuronal correlations in visual cortex. Journal of Neurophysiology 109: 940–947.
Patterson CA, Wissig SC, Kohn A (2013) Distinct effects of brief and prolonged adaptation on orientation tuning in primary visual cortex. Journal of Neuroscience 33: 532–543.
Wissig SC, Kohn A (2012) The influence of surround suppression on adaptation effects in primary visual cortex. Journal of Neurophysiology 107: 3370–3384.
Cohen MC, Kohn A (2011) Measuring and interpreting neuronal correlations. Nature Neuroscience 14: 811–819.
Peri Kurshan, Ph.D.
Assistant Professor, Dominick P. Purpura Department of Neuroscience
(neuroscience category)

Synaptic development, synaptogenesis, synapse function, molecular neuroscience, cellular neurobiology, synaptic cell biology, C. elegans, genetics, development
Studying synaptic development and function using C. elegans
Defects in the proper development and function of synapses lead to neurodevelopmental disorders such as Autism and Intellectual Disability, however the molecular mechanisms underlying these processes are still largely unknown. We use the nematode C. elegans, which has a simple and stereotyped nervous system and whose connectome has been fully mapped out, to investigate the conserved molecular mechanisms of synapse development. In particular, we study how presynaptic components including cell adhesion molecules, active zone scaffold proteins, calcium channels and synaptic vesicles arrive at the synapse and form a mature and fully functional presynaptic compartment. We combine the power of worm genetics with high resolution imaging and optical physiology readouts to elucidate the role of key molecules. These approaches have led to discoveries suggesting that the role of synaptic cell adhesion molecules such as Neurexin may be different than initially hypothesized, as we have shown that its role in presynaptic development is independent of extracellular activation and downstream of other initiating factors.
Selected Publications
Synaptogenic pathways. Kurshan PT, Shen K. Curr Opin Neurobiol. 2019 Apr 12;57:156–162. doi: 10.1016/j.conb.2019.03.005. [Epub ahead of print] Review.
g-Neurexin and Frizzled Mediate Parallel Synapse Assembly Pathways Antagonized by Receptor Endocytosis. Kurshan PT, Merrill SA, Dong Y, Ding C, Hammarlund M, Bai J, Jorgensen EM, Shen K. Neuron. 2018 Oct 10;100(1):150–166.e4. doi: 10.1016/j.neuron.2018.09.007.
Clarinet (CLA-1), a novel active zone protein required for synaptic vesicle clustering and release. Xuan Z, Manning L, Nelson J, Richmond JE, Colón-Ramos DA, Shen K, Kurshan PT. Elife. 2017 Nov 21;6. pii: e29276. doi: 10.7554/eLife.29276. Free PMC Article
Deep phenotyping unveils hidden traits and genetic relations in subtle mutants. San-Miguel A, Kurshan PT, Crane MM, Zhao Y, McGrath PT, Shen K, Lu H. Nat Commun. 2016 Nov 23;7:12990. doi: 10.1038/ncomms12990. Free PMC Article
Prevalent presence of periodic actin-spectrin-based membrane skeleton in a broad range of neuronal cell types and animal species. He J, Zhou R, Wu Z, Carrasco MA, Kurshan PT, Farley JE, Simon DJ, Wang G, Han B, Hao J, Heller E, Freeman MR, Shen K, Maniatis T, Tessier-Lavigne M, Zhuang X. Proc Natl Acad Sci U S A. 2016 May 24;113(21):6029–34. doi: 10.1073/pnas.1605707113. Epub 2016 May 9. Free PMC Article
Regulation of synaptic extracellular matrix composition is critical for proper synapse morphology. Kurshan PT, Phan AQ, Wang GJ, Crane MM, Lu H, Shen K. J Neurosci. 2014 Sep 17;34(38):12678-89. doi: 10.1523/jneurosci.1183–14.2014. Free PMC Article
Autonomous screening of C. elegans identifies genes implicated in synaptogenesis. Crane MM, Stirman JN, Ou CY, Kurshan PT, Rehg JM, Shen K, Lu H. Nat Methods. 2012 Oct;9(10):977–80. doi: 10.1038/nmeth.2141. Free PMC Article
Dendritic patterning: three-dimensional position determines dendritic avoidance capability. Kurshan PT, Shen K. Curr Biol. 2012 Mar 20;22(6):R192–4. doi: 10.1016/j.cub.2012.02.017. Preview. Free Article
Presynaptic alpha2delta-3 is required for synaptic morphogenesis independent of its Ca2+-channel functions. Kurshan PT, Oztan A, Schwarz TL. Nat Neurosci. 2009 Nov;12(11):1415–23. doi: 10.1038/nn.2417. Free PMC Article
Mutations in a Drosophila alpha2delta voltage-gated calcium channel subunit reveal a crucial synaptic function. Dickman DK*, Kurshan PT*, Schwarz TL. J Neurosci. 2008 Jan 2;28(1):31-8. doi: 10.1523/jneurosci.4498-07.2008. Free Article
A Drosophila kinesin required for synaptic bouton formation and synaptic vesicle transport. Pack-Chung E, Kurshan PT, Dickman DK, Schwarz TL. Nat Neurosci. 2007 Aug;10(8):980–9.
Saleem M. Nicola, Ph.D.
Associate Professor, Dominick P. Purpura Department of Neuroscience Associate Professor, Department of Psychiatry and Behavioral Sciences
(neuroscience category)

Neural circuits underlying reward-seeking behavior
My lab focuses on understanding the neural circuits responsible for reward-seeking and addictive behaviors. We use a systems-level approach that combines behavioral, pharmacological and electrophysiological techniques in awake, freely moving animals. We begin by identifying a hypothesis regarding the neural circuits underlying a particular behavior. For example, the nucleus accumbens (part of the ventral striatum) projects to motor output structures of the basal ganglia. The accumbens also receives input from limbic structures that have been suggested to process stimuli that predict events of consequence to the animal’s well-being. These limbic structures include the basolateral amygdala, which sends glutamatergic axons to the accumbens, and the ventral tegmental area (VTA), which sends a dopamine projection. Therefore, we hypothesized that the amygdala and VTA projections to the accumbens are part of the neural circuit that controls the animal’s response to reward-predictive stimuli.
To test this hypothesis, we designed a behavioral task that requires rats to respond, by pressing a lever, to an auditory stimulus that predicts sucrose reward. We then determined that the dopamine projection to the accumbens is required for this behavior by demonstrating that dopamine receptor antagonists microinjected directly into the animals’ nucleus accumbens caused animals to cease responding to the stimulus. We also showed that transient inactivation of the amygdala had the same effect. Next, we used multiple simultaneous single-unit recordings of neurons in the accumbens and amygdala to demonstrate that subpopulations of neurons were excited or inhibited by the reward-predictive stimulus. Finally, we established that stimulus-evoked excitations and/or inhibitions in the accumbens are required for the reward-seeking behavior instigated by the stimulus. We did this by inactivating either the dopaminergic VTA neurons or amygdala neurons while recording from accumbens neurons during the stimulus-evoked reward seeking task. Inactivation of either structure selectively abolished the firing of accumbens neurons responsive to reward-predictive stimuli. These experiments established that the convergence of the excitatory projection from the amygdala and dopaminergic projection from the VTA in the accumbens is an important part of the neural circuits that underlie stimulus-evoked reward-seeking behavior. Ongoing experiments seek to determine the nature of the information encoded by the firing of accumbens neurons driven by the amygdala and dopamine projections.
Drugs of abuse can also serve as rewards, often to the extent that drug-seeking (sometimes in response to drug-predictive stimuli) becomes excessive and harmful. A long-term goal of these experiments is to use our increasing knowledge of the neural circuits that control reward-seeking to ask how these circuits produce aberrant behavior (excessive drug-seeking) in addiction.
Selected Publications
Vega-Villar M, Horvitz JC and Nicola SM NMDA receptor-dependent plasticity in the nucleus accumbens connects reward-predictive cues to approach responses. Revision under review.
Caref K and Nicola SM (2018) Endogenous opioids in the nucleus accumbens promote approach to high-fat food in the absence of caloric need. eLife, 7:e34955.
Du Hoffmann J and Nicola SM (2014) Dopamine invigorates reward seeking by promoting cue-evoked excitation in the nucleus accumbens. J. Neurosci. 34:14349–14364. (Editor’s Featured Article)
Morrison SE and Nicola SM (2014) Neurons in the nucleus accumbens promote selection bias for nearer objects. J. Neurosci. 34:4147≠14162.
McGinty VB, Lardeux S, Taha SA, Kim JJ and Nicola SM (2013) Invigoration of reward-seeking by cue and proximity encoding in the nucleus accumbens. Neuron 78:910–922.
Nicola SM (2010) The flexible approach hypothesis: Unification of effort and cue responding hypotheses for the role of nucleus accumbens dopamine in the activation of reward-seeking behavior. J. Neurosci. 30:16585–16600.
Ambroggi F, Ishikawa A, Fields HL and Nicola SM (2008) Incentive cue encoding in the nucleus accumbens depends on basolateral amygdala inputs. Neuron 59:648–661.
Ishikawa A, Ambroggi F, Nicola SM and Fields HL (2008) Dorsomedial prefrontal cortex contribution to behavioral and nucleus accumbens neuronal responses to incentive cues. J. Neurosci. 28:5088–5098.
Yun IA, Wakabayashi KT, Fields HL and Nicola SM (2004) The ventral tegmental area is required for the behavioral and nucleus accumbens neuronal firing responses to incentive cues. J. Neurosci. 24:2923–2933.
Nicola SM, Yun IA, Wakabayashi KT and Fields HL (2004) Cue-evoked firing of nucleus accumbens neurons encodes motivational significance during a discriminative stimulus task. J. Neurophsyiol. 91:1840–1865.
José L. Peña, M.D., Ph.D.
Professor, Dominick P. Purpura Department of Neuroscience
(neuroscience category)

The owl’s brain is a showcase in Systems Neuroscience for allowing the analytical approach to how information is processed and represented in the brain. Owls exhibit a characteristic orienting response towards sound sources. This behavior is highly reproducible, the variables involved in triggering specific responses are well characterized, and the system affords progressively deeper levels of analysis. Whereas spatial selectivity of neurons in the owl’s auditory system is initially broad and ambiguous, sharp space-specificity emerges in high-order neurons. In the midbrain, a map of auditory space is computed based on differences in time and intensity of the acoustic signals that arrive at each ear. These binaural cues are processed in parallel pathways that converge where the map emerges. We have focused on regions of the brain that are crucial for this synthetic process: the neurons where the difference between the arrival times of the sound to each ear is initially detected, and the space-specific neurons that respond to sounds coming from unique directions. We found that well-defined computations, which match predictions made by studies of sound localization in humans, underlie the emergent response properties of these neurons. Thus, the owl’s brain provides a system to test models of psychoacoustics at levels from single cells to networks of neurons. Recently, we have studied why owls make systematic errors when localizing in peripheral space. We could predict these errors from looking at how space is represented in the owl’s brain. In addition, we could show how making errors in the periphery could help to localize in the front. In the future, we plan to study how information flows in the sound localization pathway using in vitro electrophysiology as well as the recording of neural activity in behaving animals.
Selected Publications
Pavao R, Sussman ES, Fischer BJ, Pena JL (2018) Anticipated ITD statistics built-in human sound localization. BioRxiv, doi: https://doi.org/10.1101/303347
Cazettes F, Fischer BJ, Beckert MV, Pena JL (2018) Emergence of an adaptive command for orienting behavior in premotor brainstem neurons of barn owls. Journal of Neuroscience, http://www.jneurosci.org/content/early/2018/07/16/JNEUROSCI.0947-18.2018
Batista G, Johnson JL, Dominguez E, Costa-Mattioli M, Pena JL (2018) Regulation of filial imprinting and structural plasticity by mTORC1 in newborn chickens. Scientific Reports, https://www.nature.com/articles/s41598-018-26479-1
Beckert MV, Pavao R, Pena JL (2017) Distinct correlation structure supporting a rate-code for sound localization in the owl’s auditory forebrain. eNeuro, https://doi.org/10.1523/ENEURO.0144–17.2017.
Batista G, Johnson JL, Dominguez E, Costa-Mattioli M, Peña JL. (2016) Translational control of auditory imprinting and structural plasticity by eIF2α. eLife. Dec 23; 5:e17197.
Fischer BJ, Peña JL (2016) Optimal nonlinear cue integration for sound localization. J Comput Neurosci. 2016 Oct 6.
Cazettes F, Fischer BJ, Peña JL (2016) Cue Reliability Represented in the Shape of Tuning Curves in the Owl’s Sound Localization System. J Neurosci. 2016 Feb 17;36(7):2101–10.
Rich D, Cazettes F, Wang Y, Peña JL, Fischer BJ (2015) Neural representation of probabilities for Bayesian inference. J Comput Neurosci. 38(2):315–23.
Cazettes F, Fischer BJ, Peña JL (2014) Spatial cue reliability drives frequency tuning in the barn Owl’s midbrain. Elife. 3:e04854.
Fontaine B, Köppl C, Peña JL (2015) Reverse correlation analysis of auditory-nerve fiber responses to broadband noise in a bird, the barn owl. J Assoc Res Otolaryngol. 16(1):101-19.
Alberto E. Pereda, M.D., Ph.D.
Professor, Dominick P. Purpura Department of Neuroscience
(neuroscience category)

The laboratory investigates the properties and dynamics of gap junction-mediated electrical transmission in the vertebrate brain. While the study of plasticity of chemical synapses has long been an area of primary interest to neuroscientists, less is known about the modifiability of electrical synapses. We investigate plastic properties of electrical synapses in teleosts (goldfish and zebrafish) and mammals. Lower vertebrates have provided with advantageous experimental models in which basic properties of electrical transmission can be more easily study. Auditory “mixed” (electrical and chemical) synaptic contacts on the teleost Mauthner cells offer the rare opportunity to correlate physiological properties with molecular composition and specific ultrastructural features of individual synapses. Electrical transmission at these terminals undergo activity-dependent potentiation and is mediated by gap junctions formed by fish homologs of connexin 36, a neuronal gap junction protein widely distributed across the mammalian brain. Our current work focuses on the mechanisms underlying activity-dependent changes in electrical synapses by investigating: i) Their functional relationship with glutamate receptors, ii) Their interaction with the dopaminergic and endocannabinoid systems, iii) The molecular mechanisms responsible for changes in the strength of electrical transmission, in particular the role of trafficking of gap junction channels and interactions with connexin-associated regulatory proteins, iv) Interactions between intrinsic membrane properties and gap junctional conductance, as a mechanism for the control of synaptic strength at electrical synapses. Thus, while focusing in the properties of electrical synapses, the research of our laboratory explores the complexity of synaptic transmission and signaling mechanisms in general.
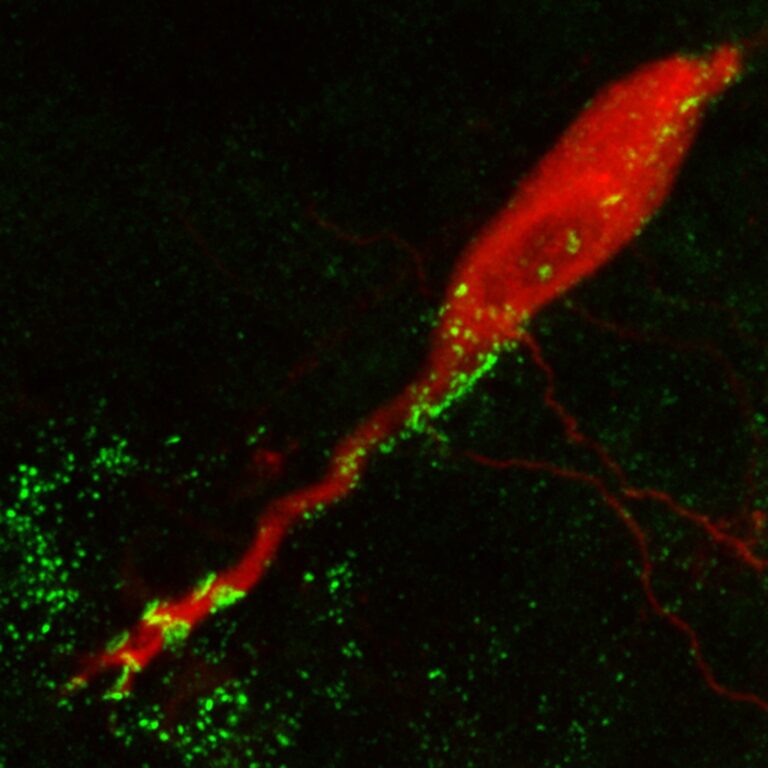
Laser-scanning confocal projection. Labeling for Cx35 (green) in a Mauthner cell of larval zebrafish backfilled with tetramethylrhodamine-dextran (red). (from Yao et al, 2014).
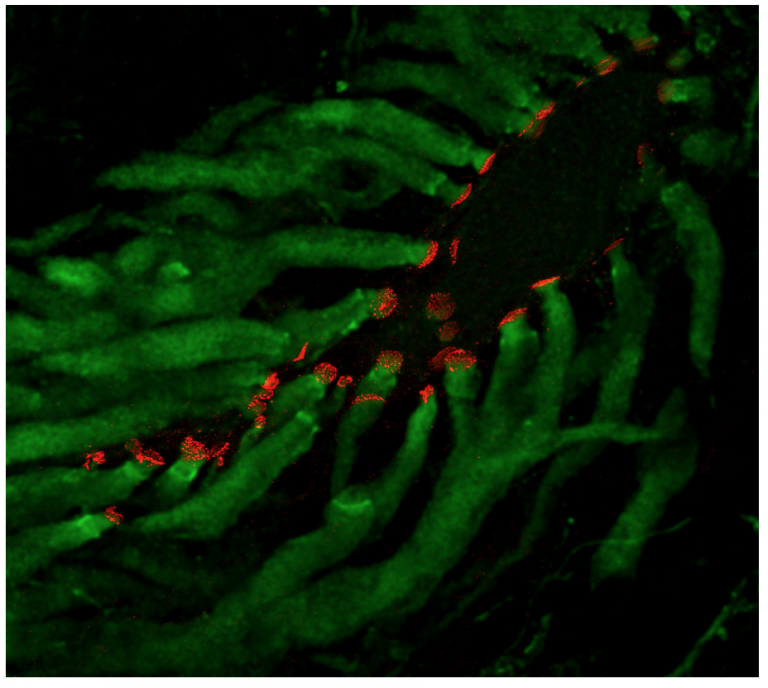
Laser-scanning confocal projection of the lateral dendrite of a goldfish Mauthner cell (unstained) illustrating saccular afferents (green) terminating as large myelinated Club endings. Labeling for Cx35 (red) reveals the areas of contact between Club endings and the dendrite. (from Flores et al., 2010)
Selected Publications
Alcamí P. and Pereda A. (2019) Beyond plasticity: the dynamic impact of electrical synapses on neural circuits, Nature Reviews Neuroscience, 20:253-271.
Marsden K.C., Jain R.A., Wolman M., Echeverry F.A., Nelson J.C., Hayer K.E., Miltenberg. B., Pereda A.E., and Granato M. A (2018) Cyfip2-dependent excitatory interneuron pathway establishes the innate startle threshold. Cell Reports, 23:878–887.
Jain R.A., Wolman M.A., Marsden K.C., Nelson J.C., Shoenhard H., Echeverry F.A., Szi C., Bell H., Skinner J., Cobbs E.N., Sawada K., Zamora A., Pereda A.E., Granato M. (2018) A forward genetic screen in zebrafish identifies the G-protein coupled receptor CaSR as a modulator of sensorimotor decision-making. Current Biology, 28:1357–1369.
Cachope R., and Pereda A. (2015) Opioids potentiate electrical synaptic transmission at mixed synapses on the Mauthner cell, Journal of Neurophysiology, 114:689–697.
Yao C., Davidson K., Delfiner M., Eddy V., Lucaci A., Soto-Riveros C., Yasumura T., Rash J.E., Pereda A. (2014) Electrical synaptic transmission in developing zebrafish: properties and molecular composition of gap junctions at a central auditory synapse, Journal of Neurophysiology, 112:2102–2113.
Pereda A. (2014) Electrical synapses and their interactions with chemical synapses. Nature Reviews Neuroscience 15:250–263.
Rash J.E., Curti S., Davidson K.G.V., Nannapaneni S., Palacios-Prado N., Flores C., Yasumura T., O’Brien J., Bukauskas F., Nagy J.I. and Pereda A. (2013) Molecular and functional asymmetry at a vertebrate electrical synapse. Neuron 79:957–969.
Curti S., Hoge G., Nagy J.I. and Pereda A. (2012) Synergy between electrical coupling and membrane properties promotes strong synchronization of neurons of the mesencephalic trigeminal nucleus. The Journal of Neuroscience 32:4341–4359.
Flores C., Nannapaneni S., Davidson K., Yasumura T., Bennett, M.V.L., Rash J.R. and Pereda A. (2012) Trafficking of gap junction channels at a vertebrate electrical synapse in vivo. Proceedings of the National Academy of Sciences (USA) 109:E573–582.
Cachope R., Mackie K., Triller A., O’Brien J. and Pereda A. (2007) Potentiation of electrical and glutamatergic synaptic transmission mediated by endocannabinoids. Neuron 56:1034–1047.
R. Suzanne Zukin, Ph.D.
Professor, Dominick P. Purpura Department of Neuroscience F.M. Kirby Chair of Neural Repair and Protection Director, Neuropsychopharmacology Center
(neuroscience category)

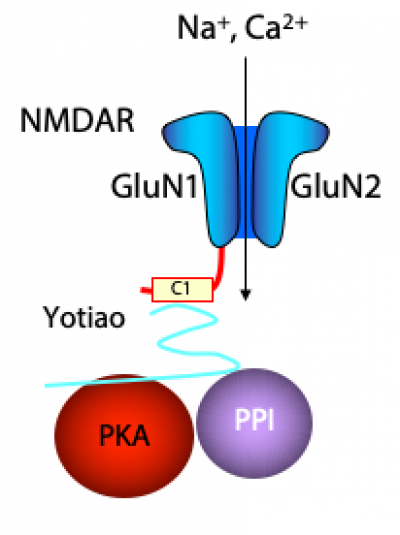
There are four major lines of ongoing research in the Zukin lab. First, we are studying the molecular and cellular mechanisms that regulate N-methyl-D-aspartate-type glutamate receptor (NMDA receptor) expression at synapses in the brain. We discovered that the switch in NMDA receptor phenotype at hippocampal synapses during normal brain development is regulated by epigenetics in an experience-dependent manner. In normal brain, the gene silencing transcription factor REST is activated during a brief window of time in differentiated neurons of the hippocampus, a brain center implicated in learning and memory, and drives the switch from immature to mature NMDA receptors. Remarkably, depriving pups of maternal access for brief periods of time during the first postnatal week prevents activation of REST and epigenetic modifications essential to acquisition of mature NMDA receptors and normal brain development. These findings have striking implications for treatment of anxiety, post-traumatic stress and other disorders associated with early maternal separation. New questions are: What is the mechanism by which REST is activated during brain development? Do other forms of stress regulate the switch in NMDA receptors? What are the consequences of blocking the switch? Our interest stems from the fact that NMDA receptors play a central role in cognitive functions such as learning and memory, synaptic plasticity and formation of neural circuitry. NMDA receptor dysregulation is implicated in Alzheimer’s disease, Huntington’s disease, AIDS dementia, stroke and schizophrenia.
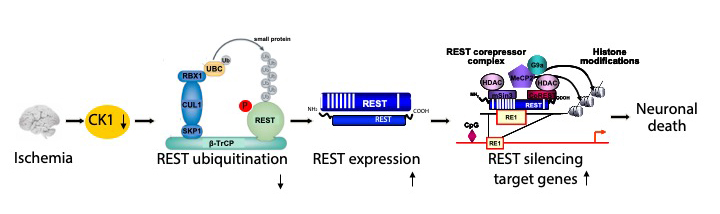
Second, we are studying the molecular and cellular mechanisms that underlie the neuronal death associated with stroke and epilepsy. We discovered that neuronal insults activate REST in selectively vulnerable adult hippocampal neurons. Upon activation, REST orchestrates epigenetic reprogramming of neuronal genes in differentiated neurons. We further showed that prolonged activation of REST is causally related to neuronal death in a clinically-relevant model of ischemic stroke. A key downstream target of REST in insulted CA1 neurons is the gene encoding the AMPA receptor subunit GluA2. This is of interest because the GluA2 subunit governs calcium permeability, channel conductance and AMPA receptor trafficking to and from synaptic sites. GluA2-lacking AMPA receptors are highly permeable to calcium and zinc, which rise to toxic levels in insulted neurons. Objectives are: 1) to understand how REST is activated in insulted neurons; 2) to examine epigenome-wide dysregulation of REST targets in stroke, Huntington’s disease and Alzheimer’s disease; and 3) to identify novel strategies to protect the human brain from neurodegeneration. Our interest stems from the known role of AMPA receptors in neuronal death arising in stroke, epilepsy, ALS and spinal cord injury.
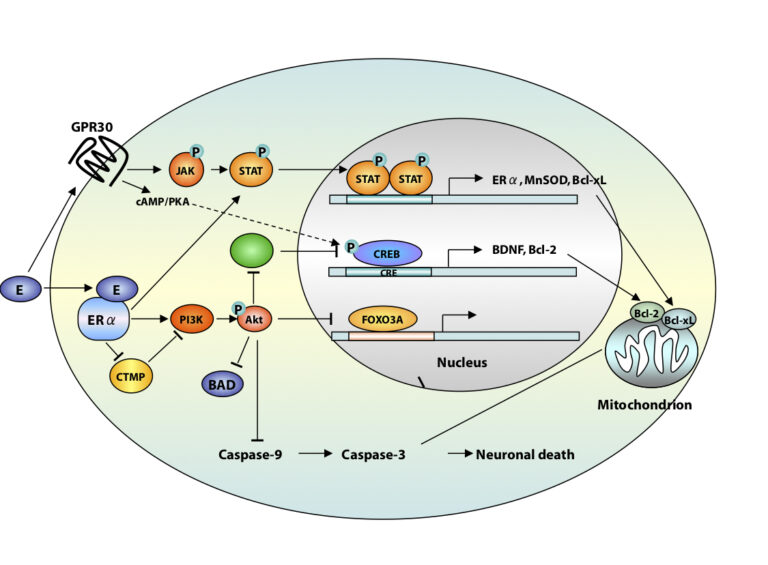
A third area of interest is that of estrogen neuroprotection in animal models of stroke, including global ischemia. Recently, we found that long-term treatment with estrogen at physiological levels ameliorates death of hippocampal neurons and cognitive deficits associated with global ischemia. We showed that ischemia and estrogen act synergistically to activate the transcription factor STAT3 and promote transcription of survivin, an inhibitor of apoptosis protein and gene target of STAT3, in insulted CA1 neurons. In experiments in which we employ direct delivery of shRNA constructs into the hippocampal CA1 of living animals, we found that STAT3 and survivin are essential to estrogen neuroprotection. These findings identify STAT3 and survivin as therapeutic targets in a clinically-relevant model of stroke. Objectives are to identify epigenetic mechanisms by which estrogen rescues neurons. Our interest stems from data that estrogen reduces the risk of cardiac arrest and stroke in animal models.
A fourth area of interest is that of RNA trafficking and targeting to dendrites and local protein synthesis in Fragile X syndrome. We found that mTOR signaling is overactivated in hippocampal neurons of Fragile X mice and causally related to aberrant synaptic plasticity. We also found that targeting of AMPAR mRNAs to synapses under basal conditions and in response to mGluR signaling is dysregulated in Fragile X neurons. We are using a combination of high resolution imaging of individual mRNA molecules (in collaboration with the Singer lab), molecular biology, and electrophysiology to examine AMPAR mRNA trafficking, local translation, synaptic plasticity and spine structure in Fragile X mice. Objectives are to identify novel signaling pathways that play a role in synaptic dysfunction. We believe that understanding the mechanisms responsible for abnormal function at the synapse will advance novel therapeutic strategies to ameliorate cognitive deficits in Fragile X syndrome and unlock doors for treating other autism spectrum disorders.
Positions for graduate students and post-doctoral fellows are available in all four areas of the laboratory’s research. Independent researchers and ideas are welcome, while well-defined and achievable projects are waiting for motivated, young investigators.
Selected Publications
(From a total of 194 peer-reviewed papers and 41 book chapters)
Gompers A, Hwang J-Y, Monday HR, Buxbaum A, Yan J, Sawicka K, Castillo PE, Singer RH, Zukin RS The memory protein CPEB3 reduces synaptic incorporation of Ca2+-permeable AMPARs and loss of anti-Hebbian LTP at synapses onto inhibitory interneurons in Fragile X mice. Nat Neurosci, in review, 2017.
Yan J, Porch WM, Court-Vazquez B, Bennett MV, Zukin RS Activation of autophagy rescues synaptic and cognitive deficits in Fragile X mice Proc. Natl. Acad. Sci. USA, 2018. PMID:30242133.
Hwang JY, Zukin RS REST, a master transcriptional regulator in neurodegenerative disease. Curr Opin Neurobiol. 48:193–200, 2018. PMID:29351877.
Pyronneau A, Qionger H, Hwang J-Y, Contractor A, Zukin RS Enhanced Rac1/cofilin signaling is critical to dendritic spine defects, synaptic dysfunction and impaired sensory perception in Fragile X Syndrome. Sci Signal 10: pii: eaan0852, 2017. PMID:29114038.
Hwang J-Y, Aromolaran KA, Zukin RS The emerging field of epigenetics in neurodegeneration and neuroprotection. Nat Rev Neurosci 18:347–361, 2017. PMID: 28515491
Hwang J-Y, Gertner MJ, Pontarelli F, Bennett MVL, Ofengeim D, Zukin RS Global ischemia induces lysosomal-mediated degradation of mTOR and activation of autophagy in hippocampal neurons destined to die. Cell Death Differ 24:317–329, 2017. PMID: 27935582
Sawicka K, Pyronneau A, Chao M, Bennett MV, Zukin RS Elevated ERK/p90 ribosomal S6 kinase activity underlies audiogenic seizure susceptibility in fragile X mice. Proc Natl Acad Sci USA 113:E6290–E6297, 2016. PMID: 27663742
Choi CH et al., Multiple drug treatments that increase cAMP signaling restore long-term memory and aberrant signaling in Fragile X syndrome models. Front Behav Neurosci 10:136–142. 2016. PMID:27445731
Puckerin A, Aromolaran KA, Chang DD, Zukin RS, Colecraft HM, Boutjdir M, Aromolaran AS. hERG 1a LQT2 C-terminus truncation mutants display hERG 1b-dependent dominant negative mechanisms. Heart Rhythm. 13:1121–30, 2016. PMID: 26775140
Huber KM, Klann E, Costa-Mattioli M, Zukin RS. Dysregulation of Mammalian Target of Rapamycin Signaling in Mouse Models of Autism. J Neurosci. 35:13836–42, 2015. PMID: 26468183
Rachel A. Ross, M.D., Ph.D.
Assistant Professor, Dominick P. Purpura Department of Neuroscience Co-primary, Assistant Professor, Department of Psychiatry and Behavioral Sciences
(neuroscience category)

Selected Publications
Ross R, Wang PY, Chari M, Lam CK, Caspi L, Ono H, Muse ED, Li X, Gutierrez-Juarez R, Light PE, Schwartz GJ, Rossetti L, Lam TK. Hypothalamic PKC regulates glucose production. Diabetes. 2008 Aug; 57(8): 2061-2065.
Ross RA, Rossetti L, Lam TKT, Schwartz GJ. Differential effects of hypothalamic long chain fatty acid infusions on suppression of hepatic glucose production. Am J Physiol Endocrinol Metab. 2010 Oct; 299(4): E633-639. Kelly DJ, Barrie MB, Ross RA, Temple BA, Moses LM, Bausch DG. Housing equity for health equity–A rights-based approach to the control of Lassa fever in post-war Sierra Leone. BMC Int Health and Hum Rights. 2013 Jan; 13(2): 1-6.
Ross RA, Mandelblat-Cerf Y, Verstegen, HMJ. Interacting neural processes of feeding, hyperactivity, stress, reward, and the utility of the activity based anorexia model of anorexia nervosa. Harvard Rev Psychiatry. 2016 Nov/Dec; 24(6):416-436. PubMed PMID: 27824637.
Cassano P, Bui E, Rogers AH, Ross R, Zeng M, Nadal-Vicens M, Mischoulon D, Baker A, Worthington J, Hoge L, Alpert J, Fava M, Wong KK, Simon NM. Inflammatory Cytokines in Major Depressive Disorder: a Rigorous Case-Control Study. Australian and New Zealand Journal of Psychiatry. June 2016.
Keenan WT*, Rupp AC*, Ross RA, Somasundaram P, Hiriyanna S, Wu Z, Badea TC, Robinson PR, Lowell BB, and Hattar S. A visual circuit uses complementary mechanisms to support transient and sustained pupil constriction. eLife 2016;10.7554/eLife.15392 PMID: 27669145
Ross RA, Foster SL, Ionescu DF. The role of chronic stress in anxious depression. Chronic Stress. 2017 Feb; 1.
Ross RA, Leon S, Madara JC, Schafer D, Fergani C, Maguire CA, Verstegen AM, Brengle E, Kong D, Herbison AE, Kaiser UB, Lowell BB, Navarro VM. PACAP neurons in the ventral premammillary nucleus regulate reproductive function in the female mouse. eLife. 2018 Jun 15;7;. pii: e35960. doi: 10.7554/eLife.35960. PubMed PMID: 29905528
Maddox SA, Hartmann J, Ross RA, Ressler KJ. Neuron. 2019 April 3; Deconstructing the gestalt: Mechanisms of fear, threat, and trauma memory encoding. Neuron. April 3, 2019. 102;60-74.
Morris LA, Kremens J, Tishelman A, Ross RA. (accepted May 2019) Depression in Turner Syndrome: A systematic review. Archives of Sexual Behavior.
Verstegen AMJ, Klymko N, Zhu L, Mathai JC, Kobayashi R, Venner A, Ross RA, VanderHorst VG, Arrigoni E, Geerling JC, Zeidel ML. (accepted June 2019). Non-Crh glutamatergic neurons in Barrington’s nucleus control micturition via glutamatergic afferents from the midbrain and hypothalamus. Current Biology.
Bui E, Hellberg SN, Hoeppner SS, Rosencrans P, Young A, Ross RA, Hoge E, Simon NM. (accepted July 2019). Circulating Levels of Oxytocin May Be Elevated in Complicated Grief: A Pilot Study. Eur J Psychotraumatol. https://doi.org/10.1080/20008198.2019.1646603.
Stephanie Rudolph, Ph.D.
Assistant Professor, Dominick P. Purpura Department of Neuroscience Co-primary, Assistant Professor, Department of Psychiatry and Behavioral Sciences
(neuroscience category)

Behavioral flexibility requires the brain to constantly adapt to environmental changes and physiological state. In response to such external and internal challenges, context-specific neuromodulators act on local and long-range circuits to orchestrate functionally and anatomically connected brain regions that ultimately control behavior. Due to its abundant connections to other parts of the brain, the cerebellum has emerged as an important structure that regulates diverse behaviors, including motor function, cognitive processes, and emotional state. Accordingly, disruption of normal cerebellar function is prevalent in psychiatric and neurodevelopmental disorders, such as schizophrenia and autism. Our laboratory is using a combination of electrophysiology, genetic approaches, imaging, and behavioral testing to better understand the mechanisms that allow the cerebellum to control behavior under physiological and pathophysiological conditions.
Selected Publications
Tsai PT, Rudolph S, Guo C, Ellegood J, Gibson JM, Schaeffer SM, Mogavero J, Lerch JP, Regehr WG, Sahin M. Sensitive Periods for Cerebellar Mediated Autistic-like Behaviors. Cell Reports. 2018; 25(2), 357–367.e4
Pieper A*, Rudolph S*, Wieser GL, Götze T, Mießner H, Yonemasu T, Yan K, Tzvetanova I, Duverge Castillo B, Bode U, Bormuth I, Wadiche JI, Schwab MH, Goebbels S. *equal contribution NeuroD2 controls inhibitory circuitry formation in the molecular layer of the cerebellum. Sci Rep. 2019 Feb 5;9(1):1448. *equal contribution
Tang JC, Rudolph S, Cepko CL. Viral Delivery of GFP-Dependent Recombinases to the Mouse Brain. Methods Mol Biol. 2017; 1642:109–126. PMID: 28815497
Tang JC, Drokhlyansky E, Etemad B, Rudolph S, Guo B, Wang S, Ellis EG, Li JZ, Cepko CL. Detection and manipulation of live antigen-expressing cells using conditionally stable nanobodies. Elife. 2016 May 20;5. pii: e15312.
Witter L*, Rudolph S*, Pressler RT, Lahlaf SI, Regehr WG. 2016 Jul 20;91(2):312–9. Purkinje Cell Collaterals Enable Output Signals from the Cerebellar Cortex to Feed Back to Purkinje Cells and Interneurons. Neuron *equal contribution
Guo C, Witter L, Rudolph S, Elliott HL, Ennis KA, Regehr WG. Purkinje Cells Directly Inhibit Granule Cells in Specialized Regions of the Cerebellar Cortex. Neuron. 2016 Sep 21;91(6):1330–41.
Rudolph S, Hull C, Regehr WG. Active Dendrites and Differential Distribution of Calcium Channels Enable Functional Compartmentalization of Golgi Cells. J Neurosci. 2015, 35(47):15492–504.
Tang JC, Rudolph S, Dhande OS, Abraira VE, Choi S, Lapan SW, Drew IR, Drokhlyansky E, Huberman AD, Regehr WG, Cepko CL. Cell type-specific manipulation with GFP-dependent Cre recombinase. Nat Neurosci. 2015, 18(9):1334–41.
Coddington LT, Rudolph S, Vande Lune P, Overstreet-Wadiche L, Wadiche JI, Spillover Activation of Inhibition Segregates Interneuronal Subpopulations in the Cerebellar Cortex. Neuron 2013, 78(6):1050–62.
Leuner K, Li W, Amaral MD, Rudolph S, Calfa G, Schuwald AM, Harteneck C, Inoue T, Pozzo-Miller L. Hyperforin modulates dendritic spine morphology in hippocampal pyramidal neurons by activating Ca(2+) -permeable TRPC6 channels. Hippocampus 2012, 23(1):40–52.
Lucas L. Sjulson, M.D., Ph.D.
Assistant Professor, Psychiatry Assistant Professor, Dominick P. Purpura Department of Neuroscience
(neuroscience category)

The long-term goal of our work is to develop novel tools and strategies for clinical neuromodulation to treat drug addiction and other neuropsychiatric disorders. To this end, efforts in the lab are focused in two primary areas:
1) Understanding dysregulation of frontolimbic circuits in drug addiction-related behaviors. Our previous work studied interactions between hippocampus and nucleus accumbens in cocaine conditioned place preference, providing the first evidence that cocaine preferentially strengthens connections arising from hippocampal place cells encoding the location where the animal received the drug. We are currently studying interactions between nucleus accumbens and prefrontal cortex in the prediction of reward value, a process whose dysregulation contributes to cravings and relapse in drug addiction. We approach these questions in rodent models using a combination of behavior, high-channel count silicon probe recordings, in vivo light field and two photon imaging, optogenetic and chemogenetic manipulations, and computational approaches.
2) Developing and validating new technologies and targets for gene-based clinical neuromodulation. Our prior work focused on validating the nucleus accumbens as a target for DREADD-based modulation in alcohol use disorders, providing a possible alternative to more invasive modalities such as lesioning or deep brain stimulation. We are currently extending these studies to other targets and disease models. In parallel, we are developing and validating improved technologies for chemogenetic and optogenetic manipulation that will be suitable for future clinical use.

Selected Publications
Sjulson L, Peyrache A, Cumpelik A, Cassataro D, Buzsáki G. Cocaine place conditioning strengthens location-specific hippocampal coupling to the nucleus accumbens. Neuron. 2018 Jun 6;98(5):926–934.e5. doi: 10.1016/j.neuron.2018.04.015.
Sjulson L, Cassataro D, DasGupta S, Miesenböck G. Cell-specific targeting of genetically encoded tools for neuroscience. Annu Rev Genet. 2016 Oct 6.
Cassataro D, Sjulson L. The use of DREADDs (Designer Receptors Exclusively Activated by Designer Drugs) in transgenic mouse behavioral models. In Thiel, G (Ed.), Designer Receptors Exclusively Activated by Designer Drugs, Neuromethods Vol 108 (2015), Humana Press.
Cassataro D, Bergfeldt D, Malekian C, Van Snellenberg JX, Thanos PK, Fishell G, Sjulson L. Reverse pharmacogenetic modulation of the nucleus accumbens reduces ethanol consumption in a limited access paradigm. Neuropsychopharmacology. 2014 Jan;39(2):283–90.
Frank Soldner, M.D.
Assistant Professor, Dominick P. Purpura Department of Neuroscience
(neuroscience category)

The main goal of my research is to apply functional genomic approaches in human cells to elucidating the molecular and cellular mechanisms of complex neurological disorders such as Alzheimer’s and Parkinson’s disease. One of the major challenges of studying human complex diseases is the lack of relevant model systems that combine known genetic elements with disease-associated phenotypic readouts. This is particularly problematic for many common medical conditions including sporadic neurodegenerative diseases, which have no well-defined genetic etiology and do not follow Mendelian inheritance patterns. Epidemiology and population genetics suggest that such sporadic diseases result from a complex interaction between multiple genetic and non-genetic (lifestyle and environmental) risk factors. And although genome wide association studies (GWASs) have identified sequence variants such as single nucleotide polymorphisms (SNPs), deletions and insertions associated with a wide variety of neurological disease, the vast majority of these risk variants have no established biological relevance to disease or clinical utility for prognosis or treatment. This complexity and our limited knowledge of the underlying genetic factors have impeded our understanding of the molecular mechanisms of many complex diseases and, more importantly, limited the development of effective therapeutics.
Three major recent innovations have fundamentally changed our ability to study human neurological diseases in a cell culture dish: (i) Reprogramming of somatic cells into human induced pluripotent stem cells (hiPSCs) to generate patient-derived disease-relevant neuronal cells, (ii) the development of genome engineering technologies such as the CRISPR/Cas9 system to modify the genome in human cells and (iii) the availability of tissue-type and disease-specific genome-scale genetic and epigenetic information. Our previous work has demonstrated that integration of population genetic and genome-wide epigenetic data combined with hiPSC and gene editing technologies now enables us to dissect the functional effects of genetic risk variants in order to study human neurological disorders in a genetically controlled and systematic manner. My lab is applying this novel experimental framework to systematically link GWAS-identified sequence variants to non-coding cis-regulatory elements and establish functional assays to connect diseases-associated risk alleles with the expression of disease-relevant effector genes and cellular phenotypes. Such disease-relevant phenotypic readouts allow us to perform unbiased chemical compound and CRISPR/Cas9-based genome-scale genetic screens to identify novel disease modifiers in human neuronal cells.
Furthermore, one of the emerging challenges in the human genetics field is to understand how genetic signals from multiple risk variants interact and collectively contribute to the development of diseases or confer susceptibility to aging and additional environmental factors. The generation of genetically defined human cellular models carrying various risk variants provide a human in vitro model system to investigate how genetic, epigenetic and environmental factors are integrated to contribute to disease development and progression.
Selected Publications
Soldner, F. & Jaenisch, R. Stem Cells, Genome Editing, and the Path to Translational Medicine. Cell, 175, 615–632 (2018)
Soldner, F., Stelzer, Y., Shivalila, C. S., Abraham, B. J., Latourelle, J. C., Barrasa, M. I., Goldmann, J., Myers, R. H., Young, R. A. & Jaenisch, R. Parkinson-associated risk variant in distal enhancer of α-synuclein modulates target gene expression. Nature 533, 95–99 (2016).
Soldner, F., Laganière, J., Cheng, A. W., Hockemeyer, D., Gao, Q., Alagappan, R., Khurana, V., Golbe, L. I., Myers, R. H., Lindquist, S., Zhang, L., Guschin, D., Fong, L. K., Vu, B. J., Meng, X., Urnov, F. D., Rebar, E. J., Gregory, P. D., Zhang, H. S. & Jaenisch, R. Generation of Isogenic Pluripotent Stem Cells Differing Exclusively at Two Early Onset Parkinson Point Mutations. Cell 146, 318–331 (2011).
Soldner, F.*, Hockemeyer, D.*, Beard, C., Gao, Q., Bell, G. W., Cook, E. G., Hargus, G., Blak, A., Cooper, O., Mitalipova, M., Isacson, O. & Jaenisch, R. Parkinson’s disease patient-derived induced pluripotent stem cells free of viral reprogramming factors. Cell 136, 964–977 (2009).
Hockemeyer, D.*, Soldner, F.*, Beard, C., Gao, Q., Mitalipova, M., Dekelver, R. C., Katibah, G. E., Amora, R., Boydston, E. A., Zeitler, B., Meng, X., Miller, J. C., Zhang, L., Rebar, E. J., Gregory, P. D., Urnov, F. D. & Jaenisch, R. Efficient targeting of expressed and silent genes in human ESCs and iPSCs using zinc-finger nucleases. Nat Biotechnol 27, 851–857 (2009).
Hockemeyer, D.*, Soldner, F.*, Cook, E. G., Gao, Q., Mitalipova, M. & Jaenisch, R. A drug-inducible system for direct reprogramming of human somatic cells to pluripotency. Cell Stem Cell 3, 346–353 (2008).
(* Equally contributing authors)
David C. Spray, Ph.D.
Professor, Dominick P. Purpura Department of Neuroscience Professor, Department of Medicine (Cardiology)
(neuroscience category)

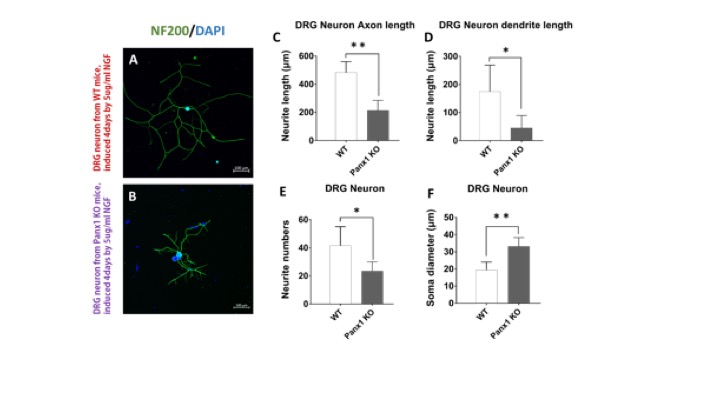
Panx1 controls DRG neuron maturation in culture. A: sensory neurons isolated from WT mice extend long processes at 4 days of NGF treatment ; B: sensory neurons isolated from Panx1 KO mice extend short processes at 4 days of NGF treatment; C,D,E: Axon and dendrite length and complexity are significantly lower in Panx1 KO mice; F: Soma diameter is significantly larger in Panx1 KO mice; (N=5; *, P<0.05; **, P< 0.01) ; Images in A and B stained with NF200 (Green) antibody and the nuclear marker DAPI (Blue)
Selected Publications
Boudewyn, L.C., Sikora, J., Kuchar, L., Ledvinova, J., Grishchuk, Y., Wang, S., Dobrenis, K., Walkley, S.U. N-butyl-deoxynojirimycin delays motor deficits, cerebellar microgliosis and Purkinje cell loss in a mouse model of mucolipidosis type IV. Neurobiol. Disease, 105:257-270, 2017.
Trilck, M., Peter, F., Zheng, C., Frank, M., Dobrenis, K., Mascher, H., Rolfs, A., Frech, M.J. Diversity of glycosphingolipid GM2 and cholesterol accumulation in NPC1 patient-specific iPSC-derived neurons. Brain Res. 1657:52-61, 2017.
Yang, D.-S., Stavrides, P., Kumar, K., Jiang, Y, Mohan, PS., Ohno, M., Dobrenis, K., Davidson, C.D., Saito, M, Pawlik, M., Huo, C., Walkley, S.U., Nixon, R.A. Cyclodextrin has conflicting actions on autophagy flux in vivo in brains of normal and Alzheimer model mice. Human Mole. Genet. 26:843-859, 2017.
Davidson, C., Fishman, YI, Puskas I., Szeman, J., Sohajda, T., McCauliff, L.A., Sikora, J., Storch, J., Vanier M.T., Szente, L., Walkley, S.U., Dobrenis, K. Efficacy and ototoxicity of different cyclodextrins in Niemann-Pick C disease. Ann Clin Transl Neurol. 3:366-380, 2016.
Saito, M., Wu, G., Hui, M., Masiello, K., Dobrenis, K., Ledeen, R.W., Saito, M. Ganglioside accumulation in activated glia in the developing brain: comparison between WT and GalNacT KO mice. J. Lipid Res. 56:1434-1448, 2015.
Farfel-Becker, T., Vitner, E.B., Kelly, S.L., Bame, J.R., Duan, J., Shinder, V., Merrill Jr, A.F., Dobrenis, K., Futerman, A.H. Neuronal accumulation of glucosylceramide in a mouse model of neuronopathic Gaucher disease leads to neurodegeneration. Human Molec. Genet 23:843-854, 2014.
Micsenyi, M.C., Sikora, J., Stephney, G., Dobrenis, K., Walkley, S.U. Lysosomal membrane permeability stimulates protein aggregate formation in neurons of a lysosomal disease. J. Neurosci. 33:10815-10827, 2013.
Erblich, B., Zhu, L., Etgen, A., Dobrenis, K., Pollard, J.W. Absence of colony stimulating factor-1 receptor signaling results in loss of microglia, disrupted brain development and olfactory deficits. PloS One, 2011. 2011;6(10):e26317. Epub 2011 Oct 27.
Stromme, P., Dobrenis, K., Sillitoe, R.V., Gulinello, M., Ali, N.F., Davidson, C., Micsenyi, M.C., Stephney, G., Ellevog, L., Klunglund, A., Walkley, S.U. X-linked Angelman-like syndrome caused by Slc9a6 knockout in mice exhibits evidence of endosomal-lysosomal dysfunction. Brain 134:3369-83, 2011.
Gulinello, M., Chen, F., Dobrenis, K. Early deficits in motor coordination, cognitive function and anxiety-like behavior in a mouse model of the neurodegenerative lysosomal storage disorder, Sandhoff disease. Behav. Brain Res. 193:315-319, 2008.
Elyse S. Sussman, Ph.D.
Professor, Dominick P. Purpura Department of Neuroscience Professor, Otorhinolaryngology—Head and Neck Surgery
(neuroscience category)

My research is in the field of Cognitive Neuroscience and is focused on under¬standing the neural bases of auditory information processing. Our laboratory’s research uses a combination of non-invasive recordings of human brain activity, in conjunction with behavioral performance measures, to specify the processes and brain structures that contribute to the organization, stor¬age and perception of a coherent sound environment. The lab investigates effects of typical and atypical neural development on auditory perception, memory, and attention across the lifespan.
Selected Publications
Symonds, R, Lee, W., Kohn, A., Schwartz, O., Witkowski, S., & Sussman, E. S. (2017). Distinguishing stimulus specific adaptation and predictive coding hypotheses in auditory change detection. Brain Topography, 30, 136–148.
Yu, Y.H., Shafer, V.L., & Sussman, E.S. (2017). Neurophysiological and behavioral responses of Mandarin lexical tone processing. Frontiers in Neuroscience, 11:95 doi: 10.3389/fnins.2017.00095
Dinces, E. and Sussman, E. (2017). Attentional resources are needed for auditory stream segregation in aging. Frontiers in Aging Neuroscience, 9:414. doi: 10.3389/fnagi.2017.00414.
Sussman, E. (2017). Auditory scene analysis: An attention perspective. Journal of Speech-Language-Hearing Research. 60(10):2989-3000. doi: 10.1044/2017_JSLHR-H-17-0041.
Costa-Faidella, J., Sussman, E.S., & Escera, C. (2017). Selective entrainment of brain oscillations drives auditory perceptual organization. Neuroimage, 159, 195–206.
Moldwin, T., Schwartz, O., & Sussman, E. (2017). Statistical learning of melodic patterns influences the brain’s response to wrong notes. Journal of Cognitive Neuroscience, 29(12):2114–2122.
Yu, Y.H., Shafer, V.L., & Sussman, E.S. (2018). The duration of auditory sensory memory for vowel processing: Evidence from neurophysiological and behavioral measures. Frontiers in Psychology-Auditory Cognitive Neuroscience. doi.org/10.3389/fpsyg.2018.00335.
Pavao R, Sussman ES, Fischer BJ, Pena JL (2018) Anticipated ITD statistics built-in human sound localization. BioRxiv, doi: https://doi.org/10.1101/303347
Brace, B., Lee, W., Cole, P., & Sussman, E. (2019). Childhood leukemia survivors exhibit deficiencies in sensory and cognitive processes, as reflected by event-related brain potentials after completion of curative chemotherapy: A preliminary investigation. Journal of Clinical and Experimental Neuropsychology, in press.
Symonds, R., Zhou, J., Cole, S., Brace, K., & Sussman, E. (2019). Cognitive resources are distributed amongst the entire auditory landscape in auditory scene analysis. Psychophysiology, in press.
Vytautas Verselis, Ph.D.
Professor, Dominick P. Purpura Department of Neuroscience
(neuroscience category)

Connexins and Syndromic Sensorineural Deafness
Our work is focused on investigating the mechanistic basis of cochlear dysfunction in syndromic deafness caused by missense mutations in the GJB2 gene that encodes the human connexin 26 (Cx26) gap junction (GJ) protein. Mutations in GJB2 are one of the most common causes of inherited, non-syndromic deafness in the human population. A subset of Cx mutations leads to syndromes in which deafness is accompanied by a heterogeneous array of cutaneous manifestations. Keratitis-Ichthyosis-Deafness (KID) syndrome is one of the more severe syndromes associated with GJB2 mutations and is characterized by profound, pre-lingual sensorineural hearing loss, vascularizing keratitis, skin lesions that can be fatal due to uncontrollable sepsis and predisposition to squamous cell carcinomas. GJs, which are formed by the docking of two, so-called hemichannels (HCs), one from each of two contacting cells, are abundant between keratinocytes and between cochlear support cells and serve as pathways for direct intercellular electrical and chemical signaling. However, it is now evident that undocked Cx26 HCs can function, thereby providing a signaling role across the plasma membrane. Our principal hypothesis is that the pathogenesis of KID syndrome is the result of a new type of channelopathy, specifically mediated by Cx26 HCs that function aberrantly leading to cell dysfunction and even cell death. We use a combination of molecular, biophysical and imaging approaches to investigate the mechanisms by which hemichannels are dysfunctional in KID syndrome. We have identified a number of aberrant HC properties including altered permeability, impaired regulation by extracellular Ca2+ and pH and shifted voltage-dependent gating. Our current focus is on altered permeability to Ca2+ and ATP, two important signaling molecules in cochlea and skin. To that end we are examining the effects of expressing hCx26 mutants in exogenous expression systems and in support cells of the Organ of Corti using cochlear tissue explants. Parallel efforts are aimed at developing a mouse model for KID syndrome using a proof-of-principle 2-plasmid, Tet-On inducible expression system in cochlea developed to express mutant GJB2 transgenes in keratinocytes. Finally we plan to screen for selective blockers of Cx26 HCs, initially using a small library of compounds enriched in known ion channel pharmacophores. Lead compounds will be followed-up through medicinal chemistry approaches to increase affinity and selectivity.
Selected Publications
Srinivas M, Verselis VK and White TW (2018). Human diseases associted with connexin mutations Biochim Biophys Acta 1860:182–201. PMCID: PMC5659969
Verselis VK (2017). Connexin hemichannels and cochlear function. Neurosci Lett. Epub ahead of print. PMID: 28917982
Sanchez HA, Slavi N, Srinivas M and Verselis VK (2016). Syndroimic deafness mutations ast Asn 14 differentially alter the open stability of Cx26 hemichannels. J Gen Physiol 148:25–42. PMCID: PMC4924935
Sanchez HA and Verselis VK (2014). Aberrant Cx26 hemichannels and keratitis-ichthyosis-deafness syndrome: Insights into syndromic hearing loss. Front Cell Neurosci 8: Article 354. PMCID: PMC4209889
Sanchez HA, Bienkowski R, Slavi N, Srinivas M and Verselis VK (2014). Altered inhibition of Cx26 hemichannels by pH and Zn2+ in the A40V mutation associated with keratitis-ichthyosis-deafness syndrome. J Biol Chem, 289:21519–32. PMCID: PMC4118113
Sanchez HA, Villone K, Srinivas M and Verselis VK (2103). The D50N mutation and syndromic deafness: Altered Cx26 hemichannel properties caused by effects on the pore and intersubunit interactions. J Gen Physiol, 142:3–22. PMCID: PMC3691445
Verselis, VK and Srinivas, M (2013). Connexin channel modulators and their mechanisms of action. Neuropharm, 75:517–24. PMCID: PMC3775990
Sanchez HA, Mese G, Srinivas M, White TW and Verselis VK. (2010). Differentialy altered Ca2+ regulation and Ca2+ permeability in Cx26 hemichannels formed by the A40V and G45E mutations that cause keratitis-ichthyosis-deafness syndrome. J Gen Physiol, 136:47–62 PMCID: PMC2894548
Verselis VK and Srinivas M (2008). Extracelular divalent cations selectively modulate loop gating, one of two intrinsic forms of voltage dependent gating in connexin hemichannels. J Gen Phys 132:315–27. PMCID: PMC2518728
Chuang CF, VanHoven MK, Fetter RD, Verselis VK and Bargmann, CI. (2007). An innexin-dependent cell network establishes stochastic left-right neuronal asymmetry in C. elegans. Cell 129: 787–99. PMID: 17512411
Steven U. Walkley, D.V.M., Ph.D.
Professor, Dominick P. Purpura Department of Neuroscience Professor, Department of Pathology Professor, The Saul R. Korey Department of Neurology Director, Rose F. Kennedy Intellectual and Developmental Disabilities Research Center, Dominick P. Purpura Department of Neuroscience
(neuroscience category)

Training in Comparative Medicine and Neuroscience provided the basis for my career interests in neurogenetic disease, particularly those disorders impacting neuronal homeostatic mechanisms and resulting in intellectual disability and related neurobehavioral abnormalities. My lab has published extensively in the area of pathogenic cascade analysis in lysosomal disease, defining key changes in neuronal structure and function as a consequence of lysosomal compromise. Current studies include: (i) the causes and consequences of ectopic dendritogenesis and neuroaxonal dystrophy, (ii) altered synaptic function underlying intellectual compromise, (iii) involvement of mTOR and TFEB/TFE3 in homeostatic dysregulation following lysosomal compromise and its impact on endosomal and autophagasomal function, and (iv) the importance of metabolite salvage in lysosomal processing.
Diseases of current focus include the lysosomal diseases Niemann-Pick types A and C, mucolipidosis IV, cystinosis, GM1 and GM2 gangliosidosis, Sanfilippo type A (MPS IIIA), Batten disorders (CLN2 and CLN3) and a newly discovered endosomal disorder known as Christianson syndrome.
My lab is also significantly involved in therapy development for genetic brain disease. We were the first to show essentially complete correction of CNS disease in the lysosomal disorder known as alpha-mannosidosis through the use of bone marrow transplantation and this treatment approach is now the standard of care for children diagnosed with this rare disorder. A disease of current focus toward therapy is Niemann-Pick type C (NPC), a fatal cholesterol-glycosphingolipid lysosomal storage disorder of children. Based on our studies of glycosphingolipid processing abnormalities in NPC disease we developed the first and presently only approved (by EMEA; FDA pending) therapy for this disorder. This is the imino sugar known as N-butyldeoxynojirimycin, or miglustat, which is a partial inhibitor of glycosphingolipid synthesis.
More recently we discovered that the FDA-approved excipient known as hydroxypropyl beta-cyclodextrin is efficacious in limiting intraneuronal accumulation of both unesterified cholesterol and glycosphingolipids, and dramatically extends the lifespan in animal models of NPC disease. Research and clinical trial development was subsequently pursued through a unique scientist/clinician/parent consortium known as SOAR (Support Of Accelerated Research) for NPC disease (SOAR-NPC) and through collaboration with TRND (Therapeutics for Rare and Neglected Diseases) and NCATS (National Center for Advancing Translational Sciences) at NIH. Phase 1 trials of this compound in NPC patients began in February, 2013, followed by the Phase 2/3 trial, with outcome analysis now in progress.
“Cerebellum of a mouse modeling the lysosomal disease late-infantile neuronal ceroid lipofuscinosis (CLN2 disease). Purkinje cells are labeled red, nuclei are blue, and aberrant aggregates of the protein p62/Sqstm1 are green.” (Matt Micsenyi and Steve Walkley)
Selected Publications
Walkley SU, Abbeduto L, Batshaw ML, Bhattacharyya A, Bookheimer SY, Christian BT, Constantino JN, de Vellis J, Doherty DA, Nelson DL, Piven J, Poduri A, Pomeroy SL, Samaco RC, Zoghbi HY, Guralnick MJ; IDDRC Directors Committee. Intellectual and Developmental Disabilities Research Centers: 50 Years of Scientific Accomplishments, Ann Neurol. 2019 Jun 17. doi: 10.1002/ana.25531. [Epub ahead of print] Review. PMID:31206741
Vite, CH, Bagel, JH, Swain, GP, Prociuk M, Sikora, J… and Walkey, SU. Intracisternal cyclodextrin prevents cerebellar dysfunction and Purkinje cell death in feline Niemann-Pick type CI disease. Sci Transl Med 2015 Feb 25;7(276):276ra26.doi:10.1126/ scitranslmed. 3010101 PMID: 25717099 PMCID: PMC4415615
Micsenyi MC, Sikora J, Stephney G, Dobrenis K, Walkley SU. Lysosomal membrane permeability stimulates protein aggregate formation in neurons of a lysosomal disease. J Neurosci 33:10815–10827, 2013. PMID: 23804102 [PubMed – in process] PMCID: PMC3693058
Lieberman AP, Puertollano R, Raben N, Slaugenhaupt S, Walkley SU, Ballabio A. Autophagy in lysosomal storage disorders. Autophagy. 2012;8(5):719–30. Epub 2012/06/01. doi: 10.4161/auto.19469. PMID: 22647656; PMCID: PMC3378416.
Strømme P, Dobrenis K, Sillitoe RV, Gulinello M, Ali NF, Davidson C, Micsenyi MC, Stephney G, Ellevog L, Klungland A, Walkley SU. X-linked Angelman-like syndrome caused by Slc9a6 knockout in mice exhibits evidence of endosomal-lysosomal dysfunction. Brain : a journal of neurology. 2011;134(Pt 11):3369–83. Epub 2011/10/04. doi: 10.1093/brain/awr250. PMID: 21964919; PMCID: PMC3212719.
McGlynn R, Dobrenis K, Walkley SU. Differential subcellular localization of cholesterol, gangliosides, and glycosaminoglycans in murine models of mucopolysaccharide storage disorders. The Journal of comparative neurology. 2004;480(4):415–26. Epub 2004/11/24. doi: 10.1002/cne.20355. PMID: 15558784.
Walkley SU. Pathogenic cascades in lysosomal disease-Why so complex? Journal of inherited metabolic disease. 2009;32(2):181–9. Epub 2009/01/09. doi: 10.1007/s10545-008-1040-5. PMID: 19130290; PMCID: PMC2682782.
Davidson CD, Ali NF, Micsenyi MC, Stephney G, Reanult S, Dobrenis K, Ory DS, Vanier MT, Walkley SU. Chronic cyclodextrin treatment of murine Niemann-Pick C disease ameliorates neuronal cholesterol and glycosphingolipid storage and disease progression. PloS one. 2009;4(9):e6951. Epub 2009/09/15. doi: 10.1371/journal.pone.0006951. PMID: 19750228; PMCID: PMC2736622.
Zervas M, Somers KL, Thrall MA, Walkley SU. Critical role for glycosphingolipids in Niemann-Pick disease type C. Current biology : CB. 2001;11(16):1283–7. Epub 2001/08/30. PMID: 11525744.
Walkley SU, Thrall MA, Dobrenis K, Huang M, March PA, Siegel DA, et al. Bone marrow transplantation corrects the enzyme defect in neurons of the central nervous system in a lysosomal storage disease. Proceedings of the National Academy of Sciences of the United States of America. 1994;91(8):2970–4. Epub 1994/04/12. PMID: 8159689; PMCID: PMC43496.
R. Suzanne Zukin, Ph.D.
Professor, Dominick P. Purpura Department of Neuroscience F.M. Kirby Chair of Neural Repair and Protection Director, Neuropsychopharmacology Center
(neuroscience category)

There are four major lines of ongoing research in the Zukin lab. First, we are studying the molecular and cellular mechanisms that regulate N-methyl-D-aspartate-type glutamate receptor (NMDA receptor) expression at synapses in the brain. We discovered that the switch in NMDA receptor phenotype at hippocampal synapses during normal brain development is regulated by epigenetics in an experience-dependent manner. In normal brain, the gene silencing transcription factor REST is activated during a brief window of time in differentiated neurons of the hippocampus, a brain center implicated in learning and memory, and drives the switch from immature to mature NMDA receptors. Remarkably, depriving pups of maternal access for brief periods of time during the first postnatal week prevents activation of REST and epigenetic modifications essential to acquisition of mature NMDA receptors and normal brain development. These findings have striking implications for treatment of anxiety, post-traumatic stress and other disorders associated with early maternal separation. New questions are: What is the mechanism by which REST is activated during brain development? Do other forms of stress regulate the switch in NMDA receptors? What are the consequences of blocking the switch? Our interest stems from the fact that NMDA receptors play a central role in cognitive functions such as learning and memory, synaptic plasticity and formation of neural circuitry. NMDA receptor dysregulation is implicated in Alzheimer’s disease, Huntington’s disease, AIDS dementia, stroke and schizophrenia.
Second, we are studying the molecular and cellular mechanisms that underlie the neuronal death associated with stroke and epilepsy. We discovered that neuronal insults activate REST in selectively vulnerable adult hippocampal neurons. Upon activation, REST orchestrates epigenetic reprogramming of neuronal genes in differentiated neurons. We further showed that prolonged activation of REST is causally related to neuronal death in a clinically-relevant model of ischemic stroke. A key downstream target of REST in insulted CA1 neurons is the gene encoding the AMPA receptor subunit GluA2. This is of interest because the GluA2 subunit governs calcium permeability, channel conductance and AMPA receptor trafficking to and from synaptic sites. GluA2-lacking AMPA receptors are highly permeable to calcium and zinc, which rise to toxic levels in insulted neurons. Objectives are: 1) to understand how REST is activated in insulted neurons; 2) to examine epigenome-wide dysregulation of REST targets in stroke, Huntington’s disease and Alzheimer’s disease; and 3) to identify novel strategies to protect the human brain from neurodegeneration. Our interest stems from the known role of AMPA receptors in neuronal death arising in stroke, epilepsy, ALS and spinal cord injury.
A third area of interest is that of estrogen neuroprotection in animal models of stroke, including global ischemia. Recently, we found that long-term treatment with estrogen at physiological levels ameliorates death of hippocampal neurons and cognitive deficits associated with global ischemia. We showed that ischemia and estrogen act synergistically to activate the transcription factor STAT3 and promote transcription of survivin, an inhibitor of apoptosis protein and gene target of STAT3, in insulted CA1 neurons. In experiments in which we employ direct delivery of shRNA constructs into the hippocampal CA1 of living animals, we found that STAT3 and survivin are essential to estrogen neuroprotection. These findings identify STAT3 and survivin as therapeutic targets in a clinically-relevant model of stroke. Objectives are to identify epigenetic mechanisms by which estrogen rescues neurons. Our interest stems from data that estrogen reduces the risk of cardiac arrest and stroke in animal models.
A fourth area of interest is that of RNA trafficking and targeting to dendrites and local protein synthesis in Fragile X syndrome. We found that mTOR signaling is overactivated in hippocampal neurons of Fragile X mice and causally related to aberrant synaptic plasticity. We also found that targeting of AMPAR mRNAs to synapses under basal conditions and in response to mGluR signaling is dysregulated in Fragile X neurons. We are using a combination of high resolution imaging of individual mRNA molecules (in collaboration with the Singer lab), molecular biology, and electrophysiology to examine AMPAR mRNA trafficking, local translation, synaptic plasticity and spine structure in Fragile X mice. Objectives are to identify novel signaling pathways that play a role in synaptic dysfunction. We believe that understanding the mechanisms responsible for abnormal function at the synapse will advance novel therapeutic strategies to ameliorate cognitive deficits in Fragile X syndrome and unlock doors for treating other autism spectrum disorders.
Positions for graduate students and post-doctoral fellows are available in all four areas of the laboratory’s research. Independent researchers and ideas are welcome, while well-defined and achievable projects are waiting for motivated, young investigators.
Selected Publications
(From a total of 194 peer-reviewed papers and 41 book chapters)
Gompers A, Hwang J-Y, Monday HR, Buxbaum A, Yan J, Sawicka K, Castillo PE, Singer RH, Zukin RS The memory protein CPEB3 reduces synaptic incorporation of Ca2+-permeable AMPARs and loss of anti-Hebbian LTP at synapses onto inhibitory interneurons in Fragile X mice. Nat Neurosci, in review, 2017.
Yan J, Porch WM, Court-Vazquez B, Bennett MV, Zukin RS Activation of autophagy rescues synaptic and cognitive deficits in Fragile X mice Proc. Natl. Acad. Sci. USA, 2018. PMID:30242133.
Hwang JY, Zukin RS REST, a master transcriptional regulator in neurodegenerative disease. Curr Opin Neurobiol. 48:193–200, 2018. PMID:29351877.
Pyronneau A, Qionger H, Hwang J-Y, Contractor A, Zukin RS Enhanced Rac1/cofilin signaling is critical to dendritic spine defects, synaptic dysfunction and impaired sensory perception in Fragile X Syndrome. Sci Signal 10: pii: eaan0852, 2017. PMID:29114038.
Hwang J-Y, Aromolaran KA, Zukin RS The emerging field of epigenetics in neurodegeneration and neuroprotection. Nat Rev Neurosci 18:347–361, 2017. PMID: 28515491
Hwang J-Y, Gertner MJ, Pontarelli F, Bennett MVL, Ofengeim D, Zukin RS Global ischemia induces lysosomal-mediated degradation of mTOR and activation of autophagy in hippocampal neurons destined to die. Cell Death Differ 24:317–329, 2017. PMID: 27935582
Sawicka K, Pyronneau A, Chao M, Bennett MV, Zukin RS Elevated ERK/p90 ribosomal S6 kinase activity underlies audiogenic seizure susceptibility in fragile X mice. Proc Natl Acad Sci USA 113:E6290–E6297, 2016. PMID: 27663742
Choi CH et al., Multiple drug treatments that increase cAMP signaling restore long-term memory and aberrant signaling in Fragile X syndrome models. Front Behav Neurosci 10:136–142. 2016. PMID:27445731
Puckerin A, Aromolaran KA, Chang DD, Zukin RS, Colecraft HM, Boutjdir M, Aromolaran AS. hERG 1a LQT2 C-terminus truncation mutants display hERG 1b-dependent dominant negative mechanisms. Heart Rhythm. 13:1121–30, 2016. PMID: 26775140
Huber KM, Klann E, Costa-Mattioli M, Zukin RS. Dysregulation of Mammalian Target of Rapamycin Signaling in Mouse Models of Autism. J Neurosci. 35:13836–42, 2015. PMID: 26468183


